STAAR Surgical Announces the EVO Implantable Lens US Clinical Trial is Underway with 1st Patient Implanted at Price Vision Gr...
February 04 2020 - 7:00AM
Business Wire
STAAR Surgical Company (NASDAQ: STAA), a leading developer,
manufacturer and marketer of implantable lenses and companion
delivery systems for the eye, today announced the first patient was
implanted in the Company’s U.S. human clinical study for its EVO
ICL family of lenses. The investigational study, “A Multicenter
Clinical Evaluation of the EVO/EVO+ Visian® Implantable Collamer®
Lens,” will be conducted at more than a dozen ophthalmology clinics
across the United States. EVO Visian ICLs are intended to treat a
wide range of refractive error, including myopia (nearsightedness)
which is the need for distance vision correction. Myopia is the
most common ocular disorder worldwide and its incidence is
increasing rapidly. An estimated 1.9 billion people globally have
myopia and this number is projected to rise by 52% by the year
2050.1
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20200204005306/en/
If you suffer from nearsightedness or
astigmatism, the EVO Investigational lens is designed to improve
your distance vision without glasses or contact lenses. (Photo:
Business Wire)
“Enrolling and implanting patients in the U.S. trial of our EVO
family of implantable Collamer lenses is the next step in opening
the U.S. market, the second largest market in the world for
refractive vision correction, to our latest generation of lenses,”
said Caren Mason, President & CEO of STAAR Surgical. “Surgeons
outside the U.S. have implanted in excess of 750,000 of our EVO
lenses in patients as the need for distance vision correction
grows. We look forward to the U.S. study of our EVO lenses
generating additional safety data on our implantable Collamer
lenses.”
Ophthalmology clinics participating in the study include
locations in the following metropolitan areas: Indianapolis,
Chicago, Dallas, Washington D.C., Philadelphia, St. Louis, San
Antonio, Kansas City, Cleveland, Honolulu, Omaha, Fargo and Salt
Lake City.
The first patient in the U.S. clinical study of EVO implantable
lenses was enrolled at Price Vision Group in Indianapolis. Francis
W. Price, Jr. MD, study investigator and founder of Price Vision
Group and the Cornea Research Foundation of America, stated, “My
team and I very much enjoy investigating new and next generation
approaches that may help patients. I am honored to be the first
member of the principal investigators group to enroll and implant a
patient with the EVO ICL in the United States. This latest version
of the Visian ICL, the EVO lens, has a central port that eliminates
the need for a peripheral iridotomy (a hole in the iris or colored
part of the eye) prior to implanting the lens. Slightly smaller in
size to a contact lens, these remarkable ICL lenses are designed to
provide vision correction and intended to remain in place as long
as the patient is satisfied with his or her vision. The lenses are
made of a very thin, soft, flexible and highly-biocompatible
Collamer material and are removable if the patient is not happy, or
develops a cataract as part of the normal aging process. Price
Vision Group already has extensive experience working with the
earlier versions of the Visian ICL that have been approved in the
U.S. since 2005. Our practice has routinely sought out
opportunities to evaluate the most promising vision correction
treatments on the horizon, and we look forward to our continued
participation in this current study of the EVO lenses.”
A list of participating clinics across the U.S. and additional
details on the clinical trial protocol, which has a primary
endpoint at six months follow-up, will be posted on
ClinicalTrials.gov under “A Multicenter Clinical Evaluation of the
EVO/EVO+ Visian® Implantable Collamer® Lens” in late February.
About STAAR Surgical
STAAR, which has been dedicated solely to ophthalmic surgery for
over 30 years, designs, develops, manufactures and markets
implantable lenses for the eye with companion delivery systems.
These lenses are intended to provide visual freedom for patients,
lessening or eliminating the reliance on eyeglasses or contact
lenses. All of these lenses are foldable, which permits the surgeon
to insert them through a small incision. STAAR’s lens used in
refractive surgery is called an Implantable Collamer® Lens or
“ICL”, which includes the EVO Visian ICL™ product line. More than
1,000,000 Visian® ICLs have been implanted to date and STAAR
markets these lenses in over 75 countries. To learn more about the
ICL go to: www.discovericl.com. Headquartered in Lake Forest, CA,
the company operates manufacturing and packaging facilities in
Aliso Viejo, CA, Monrovia, CA and Nidau, Switzerland. For more
information, please visit the Company’s website at
www.staar.com.
Forward-Looking
Statements
This news release contains forward-looking statements. These
statements include but are not limited to statements regarding the
market opportunity and potential commercial and medical
significance of the EVO Visian ICL family of products in the
market, and our ability to obtain and maintain regulatory market
approval. Forward-looking statements are based on our current
expectations or beliefs regarding future events or circumstances,
and you should not place undue reliance on these statements. Such
statements involve known and unknown risks, uncertainties,
assumptions and other factors, many of which are out of STAAR’s
control and difficult to forecast that may cause actual results to
differ materially from those that may be described or implied in
the forward-looking statements. STAAR cannot be certain about the
regulatory approval or commercial success of the Visian ICL family
of products in any market. For a discussion of certain other risks,
uncertainties and other factors affecting the statements contained
in this news release, see STAAR’s Annual Report on Form 10-K for
the year ended December 28, 2018, under the caption “Risk Factors,”
which is on file with the SEC and available in the “Investor
Information” section of STAAR’s website under the heading “SEC
Filings”. Except as required by law, STAAR assumes no, and hereby
disclaims any, obligation to update any of the foregoing or any
other forward-looking statements. STAAR nonetheless reserves the
right to make such updates from time to time by press release,
periodic report or other method of public disclosure without the
need for specific reference to this news release. No such update
shall be deemed to indicate that other statements not addressed by
such update remain correct or create an obligation to provide any
other updates.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200204005306/en/
Investors & Media Brian Moore Sr. Director, Investor,
Media Relations and Corporate Development (626) 303-7902, Ext. 3023
bmoore@staar.com
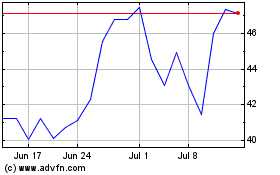
STAAR Surgical (NASDAQ:STAA)
Historical Stock Chart
From Mar 2024 to Apr 2024
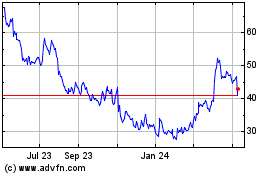
STAAR Surgical (NASDAQ:STAA)
Historical Stock Chart
From Apr 2023 to Apr 2024