Roche's New Skin Cancer Drug Zelboraf Gets European Approval
February 20 2012 - 5:01AM
Dow Jones News
Roche Holding AG (ROG.VX) Monday said European drug regulators
okayed its new skin cancer medicine Zelboraf for treating the
deadliest form of skin cancer.
European approval of the potentially super-selling drug, which
comes together with a diagnostics test that identifies melanoma
patients who may best benefit from the medicine, had been expected
and builds on the go-ahead from U.S. regulators in August 2011.
"We already included a positive European regulatory decision
scenario in our model and thus make no changes to our estimates,
and we carry peak sales for Zelboraf of CHF1.3 billion," Vontobel
pharma analyst Andrew Weiss said.
Melanoma is generally a curable disease when diagnosed early
but, when it spreads to other parts of the body, it is the
deadliest and most aggressive form of skin cancer.
A person with metastatic melanoma typically has on average a
short life expectancy that is measured in months. Only around one
in four people with metastatic melanoma are expected to be alive
one year after their diagnosis.
Zelboraf is given as a twice-daily pill and targets patients
with BRAF V600 gene mutations whose late-stage skin cancer has
spread to other parts of the body and is inoperable. Mutations of
the BRAF protein are thought to occur in around half of all
melanomas and 8% of solid cancer tumors.
Roche develops medicines together with diagnostic tests, an
approach that is aimed at helping patients and doctors establish
whether a therapeutic approach is working or not, as the tests can
signal if a patient is likely to benefit from a medicine. Roche
said the latest regulatory backing for Zelboraf is further evidence
that its strategy of developing targeted therapies is working.
"Today's approval is important news for people with BRAF
mutation-positive metastatic melanoma as Zelboraf significantly
improves patient survival and exemplifies the benefits that Roche's
personalized approach to medicine can provide for patients,
physicians and society," said Hal Barron, Roche's chief medical
officer and head of product development.
Roche, the world's biggest maker of cancer drugs, has recently
been buoyed by a more promising product pipeline.
Gene sequencing technology is a key element in the Swiss group's
oncology strategy. Its determination to acquire cutting-edge
abilities to keep advancing in that disease area lie behind Roche's
$5.7 billion bid for San Diego-based gene sequencing company
Illumina Inc. (ILMN), a leader in that field.
Roche shares at 0905 GMT were up CHF0.60, or 0.4% at CHF162.40
and 17% higher on the year, outperforming the STOXX 600 European
healthcare index, which was up 11% over from year-ago levels.
--By Sten Stovall, Dow Jones Newswires; +44 207 842 9292;
sten.stovall@dowjones.com
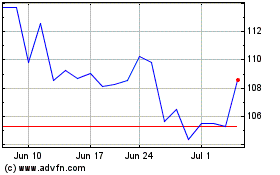
Illumina (NASDAQ:ILMN)
Historical Stock Chart
From Aug 2024 to Sep 2024
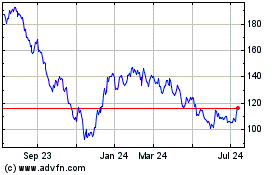
Illumina (NASDAQ:ILMN)
Historical Stock Chart
From Sep 2023 to Sep 2024