AtriCure Receives Clearance for Expanded Labeling Claims for AtriClip Devices
August 27 2019 - 8:00AM
Business Wire
Clinical evidence demonstrates that AtriClip®
devices exclude and electrically isolate the left atrial
appendage
AtriCure, Inc. (Nasdaq: ATRC), a leading innovator in surgical
treatment for atrial fibrillation (Afib) and left atrial appendage
(LAA) management, today announced that it has received U.S. Food
and Drug Administration (FDA) 510(k) clearance of additional
labeling claims for AtriClip® LAA management devices, including
changing the indication from occlusion of the LAA to exclusion, and
also adding electrical isolation as a labeling claim. Exclusion
shuts off and/or eliminates the appendage from the left atrium,
whereas occlusion plugs the opening to prevent flow into the LAA.
The electrical isolation claim was granted after testing
demonstrated that when excluding the LAA using an AtriClip device,
the appendage can no longer conduct electrical activity.
AtriClip devices are the most widely implanted left atrial
appendage management devices on the market, having been used in
more than 190,000 procedures worldwide. Over the past several
years, new iterations of the AtriClip device have been developed
and brought to market. These include enhancements to make the
devices less invasive, amenable to a wider range of patient
anatomies, and adaptable to varying operator preferences and
techniques. These new labeling claims reflect that AtriClip devices
exclude the appendage resulting in eliminating it as a source of
electrical activity through the process of ischemic necrosis. The
safety and effectiveness of AtriClip devices in rhythm control
management of an atrial arrhythmia, either alone or in combination
with ablative treatment, has not been established.
“We continue to demonstrate our leadership in the LAA management
space by expanding our labeling claims for AtriClip devices,” said
Michael Carrel, President and Chief Executive Officer of AtriCure.
“The ability to simultaneously exclude and electrically isolate the
LAA using an AtriClip device builds on our growing portfolio of
devices. Over the past several years, we have incrementally
expanded our labeling to reflect the growing clinical use for
AtriClip devices. We anticipate additional labeling expansion in
the future as the number of users of AtriClip devices grow.”
“Historically, the surgical amputation of the left atrial
appendage in a cut-and-sew fashion was considered to be the gold
standard for LAA management," said S. Patrick Whalen, MD, from Wake
Forest Baptist Health. "The AtriClip device mimics the gold
standard by eliminating the LAA by epicardial, mechanical closure.
The subsequent ischemic necrosis process ceases all electrical
activity in the LAA perioperatively and the appendage is ultimately
resorbed.”
"Our research has defined a clear benefit of the electrical
isolation of the LAA," said Dhanunjaya Lakkireddy, MD, from Kansas
City Heart Rhythm Institute (KCHRI). "The AtriClip device excludes
the left atrial appendage in a manner that could result in its
electrical isolation.”
About AtriCure, Inc.
AtriCure, Inc. provides innovative technologies for the
treatment of Afib and related conditions. Afib affects more than 33
million people worldwide. Electrophysiologists and cardiothoracic
surgeons around the globe use AtriCure technologies for the
treatment of Afib and reduction of Afib related complications.
AtriCure’s Isolator® Synergy™ Ablation System is the first and only
medical device to receive FDA approval for the treatment of
persistent Afib. AtriCure’s AtriClip® Left Atrial Appendage
Exclusion System products are the most widely sold left atrial
appendage management devices worldwide. For more information, visit
AtriCure.com or follow us on Twitter @AtriCure.
Forward-Looking Statements
This press release contains “forward-looking statements”– that
is, statements related to future events that by their nature
address matters that are uncertain. For details on the
uncertainties that may cause our actual results to be materially
different than those expressed in our forward-looking statements,
visit http://www.atricure.com/fls as well as our Annual Reports on
Form 10-K and Quarterly Reports on Form 10-Q which contain risk
factors. We do not undertake to update our forward-looking
statements. This document also includes forward-looking projected
financial information that is based on current estimates and
forecasts. Actual results could differ materially.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190827005161/en/
Valerie Storch-Willhaus Media Relations Senior Director,
Corporate Marketing and Communications (612) 605-3311
vstorch-willhaus@AtriCure.com
Andy Wade Investor Relations Senior Vice President and Chief
Financial Officer (513) 755-4564 awade@AtriCure.com
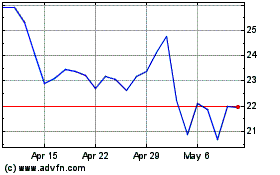
AtriCure (NASDAQ:ATRC)
Historical Stock Chart
From Mar 2024 to Apr 2024
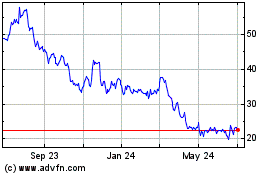
AtriCure (NASDAQ:ATRC)
Historical Stock Chart
From Apr 2023 to Apr 2024