Anika Appoints Mira Leiwant as Vice President of Regulatory Affairs, Quality, and Clinical Affairs
October 14 2019 - 8:30AM
Business Wire
Industry veteran brings extensive experience
leading global regulatory, quality, and clinical affairs teams
Anika Therapeutics, Inc. (NASDAQ: ANIK), a global, integrated
company, focused on developing regenerative therapies leveraging
its proprietary hyaluronic acid ("HA") technology platform to
provide treatments across the joint preservation and restoration
continuum of care, today announced the appointment of Mira Leiwant
to the newly created position of Vice President of Regulatory
Affairs, Quality and Clinical Affairs. Ms. Leiwant joins the
company with over 20 years of experience leading the regulatory,
quality, and clinical affairs strategies for companies specializing
in developing, manufacturing, and commercializing medical devices
and pharmaceutical products. In this role, Ms. Leiwant will oversee
Anika’s global regulatory and clinical strategy, regulatory
submissions, interactions with U.S. and international governmental
health authorities, and quality and clinical affairs teams and
processes.
“We are pleased to strengthen the executive management team’s
regulatory, quality, and clinical expertise with the addition of
Ms. Leiwant,” said Joseph Darling, President and Chief Executive
Officer of Anika Therapeutics. “Her deep experience in developing
and implementing U.S. and international regulatory strategies will
be invaluable as we advance our joint preservation and restoration
pipeline to expand our portfolio of new products in the market to
achieve our growth objectives.”
“Anika has an expansive pipeline of differentiated regenerative
therapies, and I am delighted to join its leadership team at this
transformative time for the Company,” said Mira Leiwant, Vice
President of Regulatory Affairs, Quality, and Clinical Affairs at
Anika Therapeutics. “I look forward to developing Anika’s global
regulatory and clinical strategies, as well as enhancing its
quality assurance processes to efficiently bring Anika’s innovative
solutions to patients around the world."
Prior to joining Anika, Ms. Leiwant spent three years at BTG
International, an international specialist healthcare company, as
Vice President, Regulatory Affairs. Before joining BTG
International, Ms. Leiwant served as Senior Director, Regulatory
Affairs and Quality Engineering for LifeCell Corporation, a medical
device subsidiary of Allergan. Previously, Ms. Leiwant held
leadership positions in Quality Assurance at Svelte Medical
Systems, Inc. and Vascular Therapies. Ms. Leiwant obtained both
M.E. and B.S. in Mechanical Engineering with Bioengineering option
from Cornell University.
About Anika Therapeutics, Inc.
Anika Therapeutics, Inc. (NASDAQ: ANIK) is a global,
integrated regenerative therapies company based in Bedford,
Massachusetts. Anika is committed to delivering therapies to
improve the lives of patients across a continuum of care from
osteoarthritis pain management to joint preservation and
restoration. The Company has over two decades of global expertise
commercializing more than 20 products based on its proprietary
hyaluronic acid (HA) technology platform. For more
information about Anika, please visit
www.anikatherapeutics.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20191014005026/en/
For Investor Inquiries: Anika Therapeutics, Inc. Sylvia Cheung,
781-457-9000 Chief Financial Officer
For Media Inquiries: W2O Group Sonal Vasudev, 917-523-1418,
sonal@w2ogroup.com
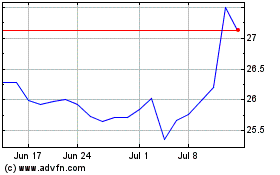
Anika Therapeutics (NASDAQ:ANIK)
Historical Stock Chart
From Mar 2024 to Apr 2024
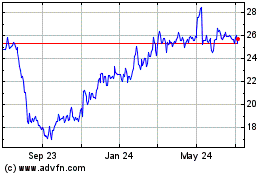
Anika Therapeutics (NASDAQ:ANIK)
Historical Stock Chart
From Apr 2023 to Apr 2024