Acutus Medical Announces Pulse Field Ablation Program and Initial Pre-Clinical Results
September 08 2020 - 8:30AM

Acutus Medical, Inc. (NASDAQ:AFIB) today announced
pre-clinical experimental results of a novel diagnostic and
therapeutic workflow utilizing the AcQMap® System along with
tissue-selective, non-thermal pulse field ablation (PFA) via a
proprietary Acutus ablation catheter. While Acutus has been known
for its rapid imaging and mapping platform along with access and
diagnostic products, the company is now aggressively pursuing the
therapeutic commercial market.
Pulsed field ablation (also known as electroporation) is an
emerging ablation modality that distinguishes itself from
traditional thermal ablation, such as radiofrequency energy
ablation and cryoablation, by delivering therapeutic energy faster
and more selectively with minimal collateral damage. While not
fully clinically proven or approved by regulatory authorities for
use in cardiac tissue in humans, recent pre-clinical and clinical
studies suggest that PFA may enable rapid targeted ablation of
cardiac muscle while leaving adjacent tissue, such as nerves, blood
vessels and the esophagus, completely unaffected. In addition, PFA
does not rely on heat or extreme cold to ablate tissue. The
application time required to create a lesion takes less than a
second compared to thermal ablation which may take many seconds to
minutes at each location.
The AcQMap cardiac mapping system, distinguishes itself from
conventional contact mapping via ultra-fast ultrasound imaging of
cardiac anatomy and information-rich display of actionable
diagnostic maps. By coupling the AcQMap System and the potential of
PFA, procedure times may be dramatically shortened while enhancing
the safety profile.
In a series of pre-clinical experiments, Acutus researchers
employed a novel workflow that involved ultrasound imaging of
whole-chamber cardiac anatomy and non-contact mapping of electrical
activity, followed by delivery of 4 to 8 contiguous ablation
lesions in regions of interest via a novel PFA catheter. Following
creation of these lesions, non-contact mapping was utilized to
assess and confirm the effectiveness of the therapy (successful
conduction block) in the treated regions. In multiple experiments
with multiple physician-advisors, this entire whole chamber map /
ablate / re-map to confirm block workflow was completed in an
average of 12 minutes. Acute ablation efficacy was further
confirmed by conventional high-density contact mapping of the
treated region with the AcQMap System and a contact mapping
catheter.
After a recovery period of 10 days, intra-cardiac re-mapping
confirmed lesion durability in the treated experimental model
indicating that the ablated regions were irreversibly treated via
PFA. Visual observation of cardiac muscle under a microscope
further confirmed that the ablated regions were clearly demarcated,
spanned the entire tissue thickness and non-cardiac tissue were not
affected. More importantly, blood vessels within and near the
ablated regions also appeared structurally unaffected.
This post study analysis indicates that durable, non-conductive
lesions were created without damage to collateral structures,
confirming the hypothesis that PFA may be tissue-selective.
“I am extraordinarily pleased with the effectiveness, speed, and
ease-of-use of this integrated system. The initial
results of the animal studies are thrilling and provide a robust
foundation for moving this program forward toward human trials. I
couldn’t be more impressed with the efforts of the
Acutus team,” said Dr. Steven Mickelsen an electrophysiologist
from the University of Iowa who was intimately involved with this
work as a clinical and lead consultant. Dr. Mickelsen is widely
regarded as the pioneer of PFA and was instrumental in bringing
this therapy to the cardiac ablation space.
“Acutus has consistently been focused on challenging industry
standards, thinking outside of the box and investing in highly
novel technologies. These initial results combining the use of our
novel mapping system with advanced pulsed field ablation technology
are quite promising,” commented Vince Burgess, President & CEO
of Acutus Medical, Inc.
Acutus Medical with its physician-advisors are preparing to
conduct clinical trials to prove the safety and efficacy of PFA in
patients with a wide range of cardiac arrhythmias.
Acutus Medical PFA Technology is not for sale in the United
States.Acutus Medical PFA Technology has not been granted CE
Mark.
About Acutus Medical Acutus Medical is a
dynamic arrhythmia care company focused on developing a full suite
of distinct, innovative technologies that provide physicians and
patients with excellent outcomes and procedural efficiency. At
Acutus, we know that seeing is better than believing. Diagnosing
and treating patients with atrial arrhythmias requires eliminating
the unknown. Acutus’ advanced cardiac imaging and mapping system
provides real-time arrhythmia visualization displaying the heart’s
true activation pattern, turning the chaos of a complex arrhythmia
into a clear vision for electrophysiologists. Founded in 2011,
Acutus is based in Carlsbad, California
US Media ContactsLevitate(260)
408-5383acutus@levitatenow.com
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/04d66ea7-e17f-41e9-b496-fe3bc2dbdb8a
https://www.globenewswire.com/NewsRoom/AttachmentNg/b3188f84-e191-4678-9944-211be7376f9c
https://www.globenewswire.com/NewsRoom/AttachmentNg/5f92b9b2-fe70-4995-a3a1-bc3de9308e58
https://www.globenewswire.com/NewsRoom/AttachmentNg/393d73c0-338a-4538-94d3-ba5907fdf91f
https://www.globenewswire.com/NewsRoom/AttachmentNg/c0f872e1-9ed6-49ca-b494-bfc67874351d
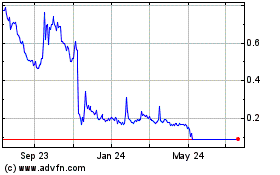
Acutus Medical (NASDAQ:AFIB)
Historical Stock Chart
From Mar 2024 to Apr 2024
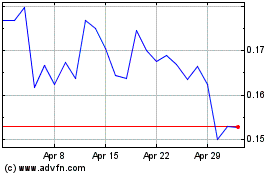
Acutus Medical (NASDAQ:AFIB)
Historical Stock Chart
From Apr 2023 to Apr 2024