130,000-square-foot Vancouver facility aims to accelerate the
development of antibody-based therapies for an array of medical
conditions.
AbCellera (Nasdaq: ABCL) today announced it has secured a site
in Vancouver for the construction of a Good Manufacturing Practices
(GMP) manufacturing facility for the production of therapeutic
antibodies, which will be the first of its kind in Canada. The
130,000-square-foot facility will expand AbCellera’s capabilities
in bringing new antibody therapies to clinical trials, supporting
Canada’s efforts to respond quickly and effectively to future
pandemics and providing AbCellera’s partners with a full solution
for accelerating their programs from a drug target to the
submission of an Investigational New Drug (IND) application.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20210622005425/en/
Rendering of AbCellera’s GMP Facility at
900-Block Evans Avenue. Images provided by Chernoff Thompson
Architects
“We envision AbCellera as a premier destination for the
brightest and most creative minds, for people who seek challenges
and who want to work at the leading edge,” said Murray McCutcheon,
Ph.D., Vice President of Corporate Development at AbCellera. “Our
recently announced headquarters and new GMP facility will empower
entirely new capabilities and jobs in Canada that we believe will
make our communities stronger, bring treatments to patients sooner,
and leave a legacy of innovation.”
This announcement follows AbCellera’s April groundbreaking on a
new global headquarters in Vancouver. The 380,000-square-foot
headquarters on 4th Avenue between Columbia and Manitoba Streets
will be within walking distance of the new GMP manufacturing
facility. The new GMP facility will be built on what is currently a
vacant two-acre site, and the planned building will house
laboratory, office, GMP protein biologic manufacturing, and
warehouse space. Design efforts are already underway and
groundbreaking is scheduled for later this year.
In parallel with the construction of the new facility, AbCellera
is also building up an internal Chemistry Manufacturing and
Controls (CMC) development organization – the team of scientists
and engineers responsible for developing the manufacturing
processes for antibody therapeutics. CMC and GMP manufacturing
capabilities will extend AbCellera’s competencies and, when
combined with AbCellera’s world-leading technology stack for
antibody discovery, will enable AbCellera to support partners to
move rapidly and seamlessly from discovery to the clinic.
“AbCellera has already redefined the speed of antibody drug
discovery, and the addition of CMC and GMP capabilities gives us
the ability to fully integrate lead therapeutic selection, moving
key choices earlier, and aligning teams to make a seamless arc of
drug development for our partners,” said Bo Barnhart, Ph.D.,
Scientific Director at AbCellera. “We are building a unique team
that includes innovative CMC Development leaders, deeply integrated
with Discovery and Translational Research, all focused on selecting
and developing better therapeutics to go from antibody discovery to
fill-finish in a year.”
The facility is scheduled to be ready for GMP manufacturing in
early 2024, and at full capacity will employ hundreds of highly
skilled scientific personnel. AbCellera anticipates adding hundreds
of employees to its current workforce of nearly 300 people over the
next few years. The development of the new GMP manufacturing
facility is funded in part by a US$125.6-million grant from the
Canadian government’s Strategic Innovation Fund (SIF) to support
AbCellera’s work and Canada’s ability to respond quickly to future
pandemics.
“I am pleased to see AbCellera participating in our government’s
effort to reverse a four-decade decline in Canada’s domestic
capacity,” said the Honourable François-Philippe Champagne,
Minister of Innovation, Science and Industry. “This is an important
step toward improving Canada’s pandemic preparedness, and a driver
for our economic recovery.”
AbCellera is at the forefront of the global COVID-19 response,
with its discovery of bamlanivimab, the world’s first monoclonal
antibody therapy for COVID-19 to be authorized for emergency use by
both the U.S. FDA and Health Canada. More than 400,000 doses of
bamlanivimab have been administered worldwide, keeping more than
22,000 patients out of hospital, and saving at least 11,000 lives.
AbCellera has also discovered a second antibody, LY-CoV1404, that
is being advanced by Eli Lilly & Company to address emerging
and future variants. This antibody has been shown to be effective
against all currently circulating variants of concern and entered
clinical trials in May.
About AbCellera Biologics Inc.
AbCellera is a technology company that searches, decodes, and
analyzes natural immune systems to find antibodies that its
partners can develop into drugs to prevent and treat disease.
AbCellera partners with drug developers of all sizes, from large
pharmaceutical to small biotechnology companies, empowering them to
move quickly, reduce costs, and tackle the toughest problems in
drug development. To learn more, please visit us at
www.abcellera.com.
AbCellera Forward-looking Statements
This press release contains forward-looking statements,
including statements made pursuant to the safe harbor provisions of
the Private Securities Litigation Reform Act of 1995. The
forward-looking statements are based on management’s beliefs and
assumptions and on information currently available to management.
All statements contained in this release other than statements of
historical fact are forward-looking statements, including
statements regarding our ability to develop, commercialize and
achieve market acceptance of our current and planned products and
services, our research and development efforts, and other matters
regarding our business strategies, use of capital, results of
operations and financial position, and plans and objectives for
future operations.
In some cases, you can identify forward-looking statements by
the words “may,” “will,” “could,” “would,” “should,” “expect,”
“intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,”
“project,” “potential,” “continue,” “ongoing” or the negative of
these terms or other comparable terminology, although not all
forward-looking statements contain these words. These statements
involve risks, uncertainties and other factors that may cause
actual results, levels of activity, performance, or achievements to
be materially different from the information expressed or implied
by these forward-looking statements. These risks, uncertainties and
other factors are described under "Risk Factors," "Management's
Discussion and Analysis of Financial Condition and Results of
Operations" and elsewhere in the documents we file with the
Securities and Exchange Commission from time to time. We caution
you that forward-looking statements are based on a combination of
facts and factors currently known by us and our projections of the
future, about which we cannot be certain. As a result, the
forward-looking statements may not prove to be accurate. The
forward-looking statements in this press release represent our
views as of the date hereof. We undertake no obligation to update
any forward-looking statements for any reason, except as required
by law.
Source: AbCellera Biologics Inc.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210622005425/en/
Inquiries Media: Jessica Yingling, Ph.D.;
media@abcellera.com, +1(236)521-6774 Business Development: Kevin
Heyries, Ph.D.; bd@abcellera.com, +1(604)559-9005 Investor
Relations: Melanie Solomon; ir@abcellera.com, +1(778)729-9116
AbCellera Biologics (NASDAQ:ABCL)
Historical Stock Chart
From Mar 2024 to Apr 2024
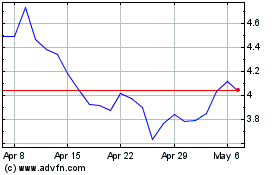
AbCellera Biologics (NASDAQ:ABCL)
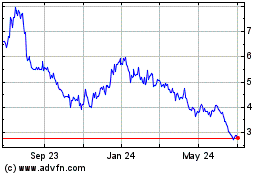
Historical Stock Chart
From Apr 2023 to Apr 2024