VBL Therapeutics Announces Appointment of Sam Backenroth as Chief Financial Officer and Establishes Operations in the United States
October 05 2021 - 8:00AM

VBL Therapeutics (Nasdaq: VBLT) today announced the appointment of
Sam Backenroth as chief financial officer (CFO) effective
immediately. Mr. Backenroth is an accomplished corporate finance
veteran with demonstrated success in building and financing private
and public biotechnology companies. In connection with the
appointment, VBL established U.S. operations in New York, which
will be managed by Mr. Backenroth, and he will be responsible for
leading the company’s future financing and business development
activities to support the company’s growth initiatives. Mr.
Backenroth succeeds Mr. Amos Ron, who is retiring but will
continue to serve as corporate secretary and in an advisory
capacity.
“On behalf of the entire board of directors and
management team, I want to thank Amos Ron for his outstanding
service to VBL, and his leadership over the past decade. We are
grateful Amos will continue as corporate secretary and an advisor
to the company,” said Prof. Dror Harats, M.D., chief executive
officer, VBL Therapeutics. “We are tremendously pleased to welcome
Sam to the VBL executive leadership team and expand our operations
in the United States. Sam has a proven track record in corporate
finance, strategy, operations and business development. His
leadership and strong investor and banking relationships will be
invaluable as we as we enter a new era of innovation and growth for
VBL as a leading global biotech company.”
Most recently, Mr. Backenroth was the chief
financial officer at NeuBase Therapeutics (Nasdaq: NBSE), a novel
genetic medicine platform company focused on rare genetic diseases
and oncology. While at NeuBase, Mr. Backenroth helped build the
finance and operations functions, took the company public, and
raised approximately $100 million in equity capital.
"I am excited to join VBL to advance the
company’s financial and business objectives and support its
continued growth and development of its first-in-class pipeline of
programs in oncology and immuno-inflammation. The company is at an
exciting point, with the ongoing Phase 3 OVAL registration-enabling
study of VB-111 in ovarian cancer advancing to the planned
progression free survival readout in the second half of 2022 and
the expected filing of an IND for the VB-601 MOSPD2 program in an
immunology application in 2022,” said Mr. Backenroth. “VBL’s gene
targeting precision medicine platform, coupled with its vision and
expertise in biology, positions the company to make meaningful
improvements to underserved patient populations and unlock
significant shareholder value. I look forward to working with the
team to capitalize on the tremendous potential for VBL to bring
novel gene therapies to people living with cancer and
immuno-inflammatory disorders.”
Prior to his tenure at NeuBase, Mr. Backenroth
was the CFO of Ohr Pharmaceutical, where he was instrumental in the
company’s growth from startup to a public market capitalization of
several hundred million and helped move its lead program from
preclinical into late-stage clinical development. He is also a
founder of Orphion Therapeutics, a company focused on one-time gene
therapy treatments for ocular and central nervous system
manifestations of ultra-rare diseases, and DepYmed, Inc., a
pharmaceutical company focused on a novel phosphatase inhibition
technology platform for rare diseases and cancer, where he
currently serves as a member of the Board of Directors. Mr.
Backenroth began his career as an investment banker with The
Benchmark Company LLC, where he raised capital and provided
advisory services for biotechnology companies. Mr. Backenroth
earned a bachelor of science degree in finance from Touro College
in New York.
VBL has a burgeoning U.S. presence at both the
board and executive level and its headquarters and manufacturing
operations will continue to be located in Israel.
About VBL TherapeuticsVascular
Biogenics Ltd., operating as VBL Therapeutics (VBL), is a late
clinical stage biopharmaceutical company focused on the discovery,
development and commercialization of first-in-class treatments for
cancer and immune or inflammatory indications. VBL has developed
three platform technologies: VTS™, a gene-therapy based platform
for targeting newly formed blood vessels with focus on cancer, an
antibody-based platform targeting MOSPD2 for anti-inflammatory and
immuno-oncology applications, and the lecinoxoids platform,
comprised of a family of small-molecules for immune-related
indications. VBL’s lead oncology product candidate, ofranergene
obadenovec (VB-111; ‘ofra-vec’), is an investigational, potentially
first-in-class, targeted anti-cancer gene-therapy agent in
development that is designed to treat a wide range of solid tumors.
VB-111 is currently being studied in a Phase 3
registration-enabling trial (NCT03398655) for recurrent
platinum-resistant ovarian cancer. To learn more about VBL
Therapeutics, please visit vblrx.com or follow the company on
LinkedIn, Twitter, YouTube or Facebook.
Forward Looking StatementsThis
press release contains forward-looking statements. All statements
other than statements of historical fact are forward-looking
statements, which are often indicated by terms such as
“anticipate,” “believe,” “could,” “estimate,” “expect,” “goal,”
“intend,” “look forward to,” “may,” “plan,” “potential,” “predict,”
“project,” “should,” “will,” “would” and similar expressions. These
forward-looking statements may include, but are not limited to,
statements regarding anticipated timing of PFS readout for the OVAL
trial, VBL’s business development efforts and financing plans as
well as its ability to bring novel gene therapies to patients.
These forward-looking statements are not promises or guarantees and
involve substantial risks and uncertainties. Among the factors that
could cause actual results to differ materially from those
described or projected herein include uncertainties associated
generally with research and development, clinical trials and
related regulatory reviews and approvals, the risk that historical
clinical trial results may not be predictive of future trial
results, that financial resources do not last for as long as
anticipated, and that VBL may not realize the expected benefits of
its intellectual property protection. In particular, the addition
of PFS as a primary endpoint in the OVAL trial is not assurance
that the trial will meet either of its primary endpoints, that it
will do so within any particular time frame, or that VBL will
obtain positive results to support any marketing application or
further development of this candidate. A further list and
description of these risks, uncertainties and other risks can be
found in VBL’s regulatory filings with the U.S. Securities and
Exchange Commission, including in its annual report on Form 20-F
for the year ended December 31, 2020, and subsequent filings with
the SEC. Existing and prospective investors are cautioned not to
place undue reliance on these forward-looking statements, which
speak only as of the date hereof. VBL undertakes no obligation to
update or revise the information contained in this press release,
whether as a result of new information, future events or
circumstances or otherwise.
CONTACT:Catherine
Day+1-917-763-2709Catherine@newdaybioconsulting.com
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/cb71632d-1aeb-4acf-9fc1-9725a13e732f
NeuBase Therapeutics (NASDAQ:NBSE)
Historical Stock Chart
From Mar 2024 to Apr 2024
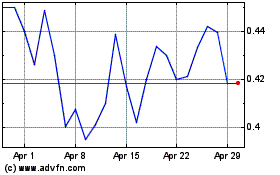
NeuBase Therapeutics (NASDAQ:NBSE)
Historical Stock Chart
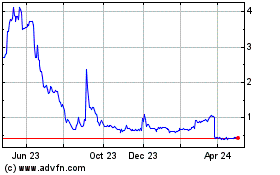
From Apr 2023 to Apr 2024