SARS-CoV-2 Spike Protein Licensed by Oragenics from the NIH Demonstrates Protective Immunity in Mice
February 02 2021 - 8:00AM
Business Wire
Data supports the Company’s approach to
COVID-19 vaccine development
Oragenics, Inc. (NYSE American: OGEN) (“Oragenics” or the
“Company”) announces that the stabilized pre-fusion spike protein
CoV-2 S-2P created by the National Institutes of Health (“NIH”) and
licensed by the Company demonstrates protective immunity in
immunized mice challenged with mouse-adapted SARS-CoV-2 virus.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20210202005441/en/
Gray dashed line = p<0.05 Gray solid
line = p<0.01 (Graphic: Business Wire)
The NIH data shows that the NIH – created COVID-19 spike protein
when dosed at either 0.1 or 1.0 mcg and combined with an adjuvant
completely inhibited virus growth in the nasal cavities and lungs
of mice when the animals were infected four weeks after their
second immunization, compared to the unvaccinated control animals.
The adjuvant uses was TLR-4-agonist Sigma Adjuvant System - a TLR-4
agonist that induces T cell activation in mice. Adjuvants are added
to vaccines to boost immune responses.
See Dinnon et al for details on the mouse-adapted challenge
model - Dinnon et al, Nature 586, 560–566 (2020) located at
- https://doi.org/10.1038/s41586-020-2708-8.
The Company previously disclosed that the NIH-produced spike
protein adjuvanted with the TLR-4-agonist Sigma Adjuvant System
generated neutralizing antibody titers, measured using a
pseudovirus neutralization assay and a plaque-reduction
neutralization titer assay. In addition, this immunization produced
a balanced Th1/Th2 (T helper cells) response. In a well-functioning
immune system both groups of T helper cells work together to keep
the system balanced.
“We are encouraged that our licensed SARS-CoV-2 spike protein
has been shown to hold promise in the creation of a COVID-19
vaccine, and believe these additional animal data supports our
continued development of Terra CoV-2, our lead COVID-19 vaccine
candidate,” said Alan Joslyn, Ph.D., President and Chief Executive
Officer of Oragenics. Dr. Joslyn continued, “Our successful pre-IND
meeting with the FDA has allowed us a clear path towards an
Investigational New Drug application. We continue to aggressively
move forward with our development strategy for this vaccine
candidate, as we believe the global need for vaccines against
SARS-CoV-2 is estimated to be between 12 billion to 14 billion
doses and the vaccines currently being administered are only a
first step towards addressing the COVID-19 pandemic.”
About Oragenics, Inc. Oragenics, Inc. is focused on the
creation of the Terra CoV-2 development of effective treatments for
novel antibiotics against infectious disease. The Company is
dedicated to the development and commercialization of a vaccine
candidate providing specific immunity from novel coronavirus. The
Terra CoV-2 immunization leverages coronavirus spike protein
research conducted by the National Institute of Health. In
addition, Oragenics has an exclusive worldwide channel
collaboration with ILH Holdings, Inc. (n/k/a Eleszto Genetika,
Inc.), relating to the development of novel lantibiotics.
Forward-Looking Statements This communication contains
“forward-looking statements” within the meaning of the safe harbor
provisions of the U.S. Private Securities Litigation Reform Act of
1995. These forward-looking statements are based on management’s
beliefs and assumptions and information currently available. The
words "believe," "expect," "anticipate," "intend," "estimate,"
"project" and similar expressions that do not relate solely to
historical matters identify forward-looking statements. Investors
should be cautious in relying on forward-looking statements because
they are subject to a variety of risks, uncertainties, and other
factors that could cause actual results to differ materially from
those expressed in any such forward-looking statements. These
factors include, but are not limited to, the following: the
Company’s ability to advance the development of Terra CoV-2 under
the timelines and in accord with the milestones it projects; the
Company’s ability to obtain funding, non-dilutive or otherwise, for
the development of Noachis Terra’s Terra CoV-2 vaccine, whether
through its own cash on hand, or another alternative source; the
regulatory application process, research and development stages,
and future clinical data and analysis relating to Terra CoV-2,
including any meetings, decisions by regulatory authorities, such
as the FDA and investigational review boards, whether favorable or
unfavorable; the potential application of Terra CoV-2 to other
coronaviruses; the Company’s ability to obtain, maintain and
enforce necessary patent and other intellectual property
protection; the nature of competition and development relating to
COVID-19 immunization and therapeutic treatments and demand for
vaccines; the Company’s expectations as to storage and
distribution, potential market and impact of other vaccines being
administered; other potential adverse impacts due to the global
COVID-19 pandemic, such as delays in regulatory review,
interruptions to manufacturers and supply chains, adverse impacts
on healthcare systems and disruption of the global economy; and
general economic and market conditions risks, as well as other
uncertainties described in our filings with the U.S. Securities and
Exchange Commission. All information set forth in this press
release is as of the date hereof. You should consider these factors
in evaluating the forward-looking statements included in this press
release and not place undue reliance on such statements. We do not
assume any obligation to publicly provide revisions or updates to
any forward-looking statements, whether as a result of new
information, future developments or otherwise, should circumstances
change, except as otherwise required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210202005441/en/
Oragenics, Inc. Michael Sullivan, CFO 813-286-7900
msullivan@oragenics.com
or
LHA Investor Relations Kim Golodetz 212-838-3777
kgolodetz@lhai.com
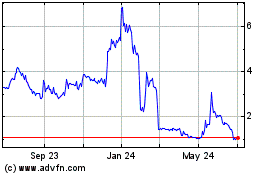
Oragenics (AMEX:OGEN)
Historical Stock Chart
From Mar 2024 to Apr 2024
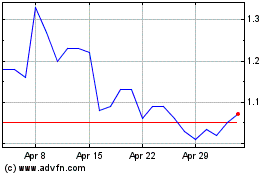
Oragenics (AMEX:OGEN)
Historical Stock Chart
From Apr 2023 to Apr 2024