Genprex Appoints William E. Gannon, Jr., MD, MBA, as Vice President of Regulatory Affairs
September 02 2020 - 8:17AM
Business Wire
Dr. Gannon will oversee and manage regulatory
matters for the Company’s upcoming clinical trials, including its
lead drug candidate in combination with Tagrisso® for which Fast
Track Designation was received earlier this year
Genprex, Inc. (“Genprex” or the “Company”) (NASDAQ: GNPX), a
clinical-stage gene therapy company developing potentially
life-changing technologies for patients with cancer and diabetes,
today announced the appointment of William Gannon, MD, MBA as the
Company’s Vice President of Regulatory Affairs. Dr. Gannon brings
more than 30 years of experience in the biotech and pharmaceutical
industries to the Company, with expertise in clinical development,
regulatory affairs and commercialization of products, and a strong
background in oncology and gene therapy.
“We are pleased to have Dr. Gannon join our team. His extensive
experience in the management of clinical trials, from designing
trials to building operational teams, will be an important asset
for us as we work toward initiating our upcoming trials and seeing
them through to successful completion,” said Rodney Varner,
Genprex’s Chairman and Chief Executive Officer.
Dr. Gannon will be responsible for managing the Company’s
regulatory affairs for its upcoming clinical trials that combine
its lead drug candidate, GPX-001 for non-small cell lung cancer
(NSCLC), with AstraZeneca’s targeted therapy Tagrisso®
(osimertinib) and with immunotherapy Keytruda® (pembrolizumab).
Genprex received Fast Track Designation from the U.S. Food and Drug
Administration (FDA) for use of GPX-001 in combination with
Tagrisso in late-stage lung cancer patients with EGFR mutations
whose tumors progress on Tagrisso. Alongside his responsibility of
advising on regulatory matters, Dr. Gannon will also serve as the
Medical Monitor, interfacing with medical personnel at trial sites
and assisting with FDA communication throughout the clinical
trials.
Dr. Gannon currently serves as Chief Scientific Officer and
Medical Director at Capital City Technical Consulting, where he has
led clinical trial development and operations management for
various clients across the biotech and pharmaceutical industries
and through which he will carry out his new Genprex role. Prior to
Capital City, Dr. Gannon served as Vice President of Clinical and
Medical Affairs for Celsion Corporation, an oncology biotech and
medical device firm, where he led its clinical trial development
and operations, data management, regulatory affairs and FDA
submissions. He has held several executive roles at various
biotech, pharmaceutical and medical device companies, overseeing
regulatory affairs, both U.S. and international, for filings,
meetings, submissions and approvals.
He has served on the Institutional Review Board (IRB), Board of
Directors and Clinical Advisory Board of several organizations and
is an active member of research and professional affiliations,
including the AdvaMed (Advance Medical Technology Association), the
Drug Information Association, the Regulatory Affairs Professional
Society and the American Academy of Pharmaceutical Physicians and
Investigators.
About Genprex, Inc.
Genprex, Inc. is a clinical-stage gene therapy company
developing potentially life-changing technologies for patients with
cancer and diabetes. Genprex’s technologies are designed to
administer disease-fighting genes to provide new treatment options
for large patient populations with cancer and diabetes who
currently have limited treatment options. Genprex works with
world-class institutions and collaborators to develop drug
candidates to further its pipeline of gene therapies in order to
provide novel treatment approaches. The Company’s lead product
candidate, GPX-001 (quaratusugene ozeplasmid), is being evaluated
as a treatment for non-small cell lung cancer (NSCLC). GPX-001 has
a multimodal mechanism of action that has been shown to interrupt
cell signaling pathways that cause replication and proliferation of
cancer cells; re-establish pathways for apoptosis, or programmed
cell death, in cancer cells; and modulate the immune response
against cancer cells. GPX-001 has also been shown to block
mechanisms that create drug resistance. In January 2020, the U.S.
Food and Drug Administration granted Fast Track Designation for
GPX-001 for NSCLC in combination therapy with osimertinib
(AstraZeneca’s Tagrisso®) for patients with EFGR mutations whose
tumors progressed after treatment with osimertinib alone. For more
information, please visit the Company’s web site at www.genprex.com
or follow Genprex on Twitter, Facebook and LinkedIn.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are "forward-looking statements"
within the meaning of the Private Securities Litigation Reform Act
of 1995. Because such statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Such
statements include, but are not limited to, statements regarding
the effect of Genprex’s product candidates, alone and in
combination with other therapies, on cancer and diabetes, regarding
potential, current and planned clinical trials, regarding the
Company’s future growth and financial status and regarding our
commercial partnerships and intellectual property licenses. Risks
that contribute to the uncertain nature of the forward-looking
statements include the presence and level of the effect of our
product candidates, alone and in combination with other therapies,
on cancer; the timing and success of our clinical trials and
planned clinical trials of GPX-001, alone and in combination with
targeted therapies and/or immunotherapies, and whether our other
potential product candidates, including GPX-002, our gene therapy
in diabetes, advance into clinical trials; the success of our
strategic partnerships, including those relating to manufacturing
of our product candidates; the timing and success at all of
obtaining FDA approval of GPX-001 and our other potential product
candidates including whether we receive fast track or similar
regulatory designations; costs associated with developing our
product candidates and whether patents will ever be issued under
patent applications that are the subject of our license agreements.
These and other risks and uncertainties are described more fully
under the caption “Risk Factors” and elsewhere in our filings and
reports with the United States Securities and Exchange Commission.
All forward-looking statements contained in this press release
speak only as of the date on which they were made. We undertake no
obligation to update such statements to reflect events that occur
or circumstances that exist after the date on which they were
made.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200902005202/en/
Genprex, Inc. (877) 774-GNPX (4679)
Investor Relations GNPX Investor Relations (877) 774-GNPX
(4679) ext. #2 investors@genprex.com
Media Contact Genprex Media Relations Kalyn Dabbs (877)
774-GNPX (4679) ext. #3 media@genprex.com
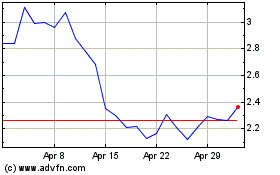
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Mar 2024 to Apr 2024
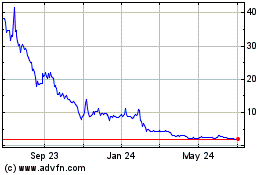
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Apr 2023 to Apr 2024