- Approval supported by efficacy and safety data from the
pivotal Phase 3 SELECT rheumatoid arthritis (RA) program, one of
the largest registrational Phase 3 programs in RA with
approximately 4,400 patients evaluated across five
studies1-5
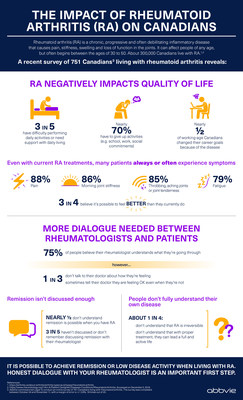
- About one in every 100 adult Canadians, or approximately
300,000 people, are living with rheumatoid arthritis, the majority
of whom don't achieve remission6,7
MONTREAL, Jan. 7, 2020 /CNW/ - AbbVie (NYSE: ABBV), a
research-based global biopharmaceutical company, today announced
that Health Canada has approved RINVOQ® (upadacitinib)
for the treatment of adults with moderately to severely active
rheumatoid arthritis who have had an inadequate response or
intolerance to methotrexate (MTX). RINVOQ is a 15 mg, once-daily
oral Janus kinase (JAK) inhibitor and may be used as monotherapy or
in combination with MTX or other nonbiologic disease-modifying
anti-rheumatic drugs (DMARDs).
"While there has been tremendous progress in the treatment of
rheumatoid arthritis over the past two decades, too many patients
still don't reach remission and continue to suffer from pain,
fatigue and morning joint stiffness," said Dr. Edward Keystone, MD,
FRCPC, Professor of Medicine, University of
Toronto. "RINVOQ had one of the largest Phase 3 clinical
trial programs in rheumatoid arthritis, and this medicine has the
potential to significantly improve signs and symptoms of the
disease."
Gaétane Lepire, who lives with rheumatoid arthritis, explained
the impact the disease has had on her life: "It affected me because
I had pain in my feet, my knees, my back and my hands 24 hours a
day, seven days a week. I tried to function as if everything were
okay, but it affected me, and sometimes I couldn't do the things I
was used to doing. I had trouble getting out of bed, going up and
down three steps, getting in and out of the car, etc."
"Rheumatoid arthritis affects 1 in 100 Canadians and causes
disability in many. RINVOQ has been approved for the treatment of
moderate to severe active rheumatoid arthritis and offers hope for
patients with this destructive autoimmune disease," explained
Dr. Janet Pope, MD, MPH, FRCPC,
Professor of Medicine, Division of Rheumatology, the Schulich
School of Medicine & Dentistry at Western
University, and Medical Director of the Rheumatology Centre
at St. Joseph's Health Care
London.
Designed to help accommodate the physical limitations of people
living with RA, the packaging for RINVOQ includes a bottle cap with
a wide, easy-to-grip texture and an embedded tool that punctures
the foil liner to simplify medication access.
"For many people living with arthritis, the act of opening a
medicine bottle or picking up a tablet can be extremely difficult,
and AbbVie designed the RINVOQ tablet, bottle and cap with this in
mind," said Ken Gagnon, Vice
President, Corporate Partnerships, The Arthritis Society. "We are
pleased to give this medication our Ease of Use designation, which
recognizes products that are designed to make life easier for
people with arthritis."
"AbbVie has been dedicated to discovering and delivering
innovative therapies for people living with rheumatic diseases for
nearly two decades," added Stéphane Lassignardie, Vice President
and General Manager, AbbVie Canada. "We are proud to expand our
portfolio of treatment options for Canadians living with RA,
particularly by offering people the convenience of a medication
taken orally, in a once-daily extended-release tablet."
About the Phase 3 SELECT Rheumatoid Arthritis Program
The Health Canada approval of RINVOQ was supported by data from
the global Phase 3 SELECT rheumatoid arthritis program, which
evaluated nearly 4,400 patients with moderate to severe active
rheumatoid arthritis in five pivotal
studies.1-5
The studies include assessments of efficacy, safety and
tolerability across a variety of patients, including those who were
methotrexate naïve or had an inadequate response to methotrexate
(MTX), patients who were intolerant to conventional synthetic
disease-modifying anti-rheumatic drugs (csDMARDs), and patients who
failed or were intolerant to biologic disease-modifying
anti-rheumatic drugs (DMARDS).1-5
- SELECT-EARLY was a 48-week trial in 947 patients with
moderately to severely active rheumatoid arthritis who were naïve
to methotrexate.
- SELECT-MONOTHERAPY was a 14-week monotherapy trial in 648
patients with moderately to severely active rheumatoid arthritis
who had an inadequate response to methotrexate.
- SELECT-NEXT was a 12-week trial in 661 patients with moderately
to severely active rheumatoid arthritis who had an inadequate
response to conventional synthetic disease modifying anti-rheumatic
drugs.
- SELECT-COMPARE was a 48-week trial in 1,629 patients with
moderately to severely active rheumatoid arthritis who had an
inadequate response to methotrexate.
- SELECT-BEYOND was a 12-week trial in 499 patients with
moderately to severely active rheumatoid arthritis who had an
inadequate response or intolerance to biologic disease-modifying
anti-rheumatic drugs.
About RINVOQ®
(upadacitinib)8
RINVOQ® (upadacitinib)
is a 15 mg, once-daily oral medication in an extended-release
tablet. It is a Janus kinase (JAK) inhibitor that interferes with
the JAK-STAT signaling pathway, which is thought to play a role in
inflammatory response.
RINVOQ is indicated for the treatment of adults with moderately
to severely active rheumatoid arthritis who have had an inadequate
response or intolerance to methotrexate. RINVOQ may be used as
monotherapy or in combination with other nonbiologic DMARDs.
For important safety information, please consult the RINVOQ
Product Monograph at www.abbvie.ca.
About AbbVie Care
Canadians prescribed RINVOQ will
have the opportunity to be enrolled in AbbVie Care, AbbVie's
signature care program. The program is designed to provide a wide
range of customized services such as reimbursement and financial
support, pharmacy services, lab work reminders and coordination, as
well as personalized education and ongoing disease management
support from a dedicated AbbVie Care nurse, throughout the
treatment journey. For more information, consult
www.abbviecare.ca.
About AbbVie
AbbVie is a global, research and
development-based biopharmaceutical company committed to developing
innovative advanced therapies for some of the world's most complex
and critical conditions. The company's mission is to use its
expertise, dedicated people and unique approach to innovation to
markedly improve treatments across four primary therapeutic areas:
immunology, oncology, virology and neuroscience. In more than
75 countries, AbbVie employees are working every day to advance
health solutions for people around the world. For more information
about AbbVie, please visit us at www.abbvie.ca and
www.abbvie.com. Follow @abbviecanada and @abbvie on
Twitter or view careers on our Facebook or
LinkedIn page.
References:
|
1
|
Genovese MC, et al.
Safety and efficacy of upadacitinib in patients with active
rheumatoid arthritis refractory to biologic disease-modifying
anti-rheumatic drugs (SELECT-BEYOND): a double-blind, randomised
controlled phase 3 trial. Lancet. 2018 Jun 23;391(10139):2513-2524.
doi: 10.1016/S0140-6736(18)31116-4. Epub 2018 Jun 13
|
|
|
|
2
|
Smolen JS, et al.
Upadacitinib as monotherapy in patients with active rheumatoid
arthritis and inadequate response to methotrexate
(SELECT-MONOTHERAPY): a randomised, placebo-controlled,
double-blind phase 3 study. Lancet. 2019. May 23. doi:
10.1016/S0140-6736(19)30419-2. Epub 2019 May 23
|
|
|
|
3
|
Fleischmann R, et al.
Upadacitinib versus placebo or adalimumab in rheumatoid arthritis
and an inadequate response to methotrexate: Results of a Phase 3,
Double-Blind, Randomized Controlled Trial. Arthritis and
Rheumatology. 2019. Jul 9. doi: 10.1002/art.41032
|
|
|
|
4
|
van Vollenhoven R, et
al. A Phase 3, Randomized, Controlled Trial Comparing Upadacitinib
Monotherapy to MTX Monotherapy in MTX-Naïve Patients with Active
Rheumatoid Arthritis. 2018 ACR/ARHP Annual Meeting; 891
|
|
|
|
5
|
Cohen S, et al.
Safety profile of upadacitinib in Rheumatoid Arthritis: Integrated
analysis from the SELECT Phase 3 Clinical Program. EULAR 2019;
THU0167
|
|
|
|
6
|
Arthritis Society,
What is rheumatoid arthritis. Available at:
https://arthritis.ca/about-arthritis/arthritis-types-(a-z)/types/rheumatoid-arthritis,
Accessed December 11, 2019
|
|
|
|
7
|
Ajeganova S. and
Huizinga T. Sustained remission in rheumatoid arthritis: latest
evidence and clinical considerations. Ther Adv Musculoskelet Dis.
2017 Oct;9(10):249-262. doi: 10.1177/1759720X17720366
|
|
|
|
8
|
RINVOQ Product
Monograph. Available at:
http://www.abbvie.ca/content/dam/abbviecorp/ca/en/docs/RINVOQ_PM.pdf.
Accessed January 6, 2020
|
SOURCE AbbVie Canada