Genmab: EC Approves Expanded Darzalex Use in Multiple Myeloma
November 19 2019 - 11:55AM
Dow Jones News
By Colin Kellaher
Genmab A/S (GMAB.KO, GMAB) on Tuesday said the European
Commission approved the expanded use of Darzalex in a form of the
blood cancer multiple myeloma.
The Copenhagen biotechnology company said the EC granted
marketing authorization for Darzalex in combination with
lenalidomide and dexamethasone for adults with newly diagnosed
multiple myeloma who are ineligible for autologous stem-cell
transplant.
Johnson & Johnson's (JNJ) Janssen Biotech Inc. unit has an
exclusive license to develop, manufacture and commercialize
Darzalex under an August 2012 agreement with Genmab.
The European Medicines Agency's Committee for Medicinal Products
for Human Use last month recommended approval of the additional
indication.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
November 19, 2019 11:40 ET (16:40 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
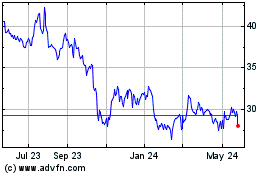
Genmab AS (NASDAQ:GMAB)
Historical Stock Chart
From Mar 2024 to Apr 2024
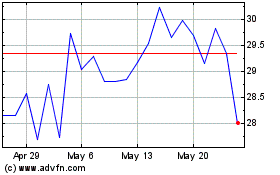
Genmab AS (NASDAQ:GMAB)
Historical Stock Chart
From Apr 2023 to Apr 2024