ROCK HILL, S.C., Sept. 12, 2019 /PRNewswire/ -- 3D Systems
(NYSE:DDD) announced today it received additional 510(k) clearance
for its D2P™ (DICOM-to-PRINT) software allowing clinicians to 3D
print diagnostic patient-specific anatomic models. 3D Systems is
the only company to offer a solution that combines its own software
and printers to create 3D printed patient-specific anatomic models
for diagnostic purposes in an unmatched breadth of medical
specialties including: cardiovascular, craniofacial,
gastrointestinal, genitourinary, neurological, and musculoskeletal
applications. D2P relies on unique automatic segmentation tools
driven by deep learning that allow medical practitioners to quickly
create accurate, digital 3D anatomic models from medical imaging
data. With the additional FDA clearance, D2P addresses the growing
demand by point of care (POC) institutions for in-house
manufacturing using an accurate and reliable 3D segmentation
solution that can produce 3D printed models.
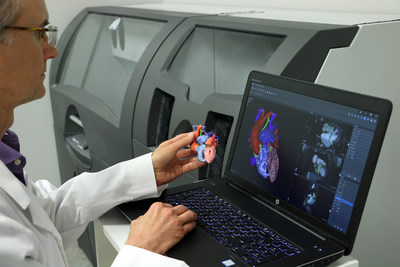
D2P now also includes the latest advancements in deep learning
image processing technology and virtual reality visualization
allowing hospitals and device manufacturers to significantly reduce
the time associated with the creation of 3D models.
The software also includes a volumetric VR solution enabling
instant views of patient scans in a 3D environment - facilitating
surgical planning and conversations between medical staff and their
patients.
"We are used to going into surgery with uncertainties and an
arsenal of contingency plans," said Dr. Solomon Dadia, deputy director of the
orthopedic-oncology department and director of the 3D surgical
center at Souraski Medical Center in Tel-Aviv. "With 3D printed models and enhanced
3D visualization tools such as VR, we are able to gain a better
understanding of the surgery and pathology we are going to treat.
This allows us to come up with a more precise surgery plan designed
to deliver a better surgical outcome."
In accordance with the FDA announcement on new guidelines for 3D
printed patient-specific anatomic models in 2017, diagnostic
quality models must be an output of a Class II regulated medical
device software. 3D Systems is the only company to offer both a
software solution and compatible printers of its own that meet this
regulatory requirement. Anatomic models can be produced using a
variety of 3D Systems printing technologies - ColorJet Printing,
MultiJet Printing, Stereolithography, and Selective Laser Sintering
– including materials that are capable of sterility and
biocompatibility.
"The capabilities offered in D2P give the healthcare
professional an unprecedented toolset for deeper medical
understanding across most medical specialties," said Ran Bronstein,
vice president, chief research and operation officer, 3D
Systems. "Our proprietary advanced visualization technology is
changing how medical imaging data is used in a variety of formats
such as virtual reality and 3D printed physical models."
To learn more about 3D Systems' solutions, please visit the 3D
Systems website.
Forward-Looking Statements
Certain statements made in
this release that are not statements of historical or current facts
are forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. Forward-looking
statements involve known and unknown risks, uncertainties and other
factors that may cause the actual results, performance or
achievements of the company to be materially different from
historical results or from any future results or projections
expressed or implied by such forward-looking statements. In many
cases, forward-looking statements can be identified by terms such
as "believes," "belief," "expects," "may," "will," "estimates,"
"intends," "anticipates" or "plans" or the negative of these terms
or other comparable terminology. Forward-looking statements are
based upon management's beliefs, assumptions, and current
expectations and may include comments as to the company's beliefs
and expectations as to future events and trends affecting its
business and are necessarily subject to uncertainties, many of
which are outside the control of the company. The factors described
under the headings "Forward-Looking Statements" and "Risk Factors"
in the company's periodic filings with the Securities and Exchange
Commission, as well as other factors, could cause actual results to
differ materially from those reflected or predicted in
forward-looking statements. Although management believes that the
expectations reflected in the forward-looking statements are
reasonable, forward-looking statements are not, and should not be
relied upon as a guarantee of future performance or results, nor
will they necessarily prove to be accurate indications of the times
at which such performance or results will be achieved. The
forward-looking statements included are made only as of the date of
the statement. 3D Systems undertakes no obligation to update or
review any forward-looking statements made by management or on its
behalf, whether as a result of future developments, subsequent
events or circumstances or otherwise.
About 3D Systems
More than 30 years ago, 3D Systems
brought the innovation of 3D printing to the manufacturing
industry. Today, as the leading AM solutions company, it empowers
manufacturers to create products and business models never before
possible through transformed workflows. This is achieved with the
Company's best-of-breed digital manufacturing ecosystem - comprised
of plastic and metal 3D printers, print materials, on-demand
manufacturing services and a portfolio of end-to-end manufacturing
software. Each solution is powered by the expertise of the
company's application engineers who collaborate with customers to
transform manufacturing environments. 3D Systems' solutions address
a variety of advanced applications for prototyping through
production in markets such as aerospace, automotive, medical,
dental and consumer goods. More information on the company is
available at www.3dsystems.com.

 View original content to download
multimedia:http://www.prnewswire.com/news-releases/3d-systems-draws-on-healthcare-expertise-to-deliver-fda-cleared-d2p--industrys-only-company-to-create-patient-specific-diagnostic-anatomic-models-using-its-own-software-and-printers-300917207.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/3d-systems-draws-on-healthcare-expertise-to-deliver-fda-cleared-d2p--industrys-only-company-to-create-patient-specific-diagnostic-anatomic-models-using-its-own-software-and-printers-300917207.html
SOURCE 3D Systems