FDA Approves Philip Morris International Heat-Not-Burn IQOS Device for Sale in U.S.
April 30 2019 - 3:00PM
Dow Jones News
By Jennifer Maloney
The Food and Drug Administration said it would allow two of the
world's biggest cigarette makers to start selling in the U.S. a
hand-held device that heats but doesn't burn tobacco, though the
agency hasn't yet ruled on whether they will be allowed to market
it as safer than cigarettes.
Philip Morris International Inc. has spent billions of dollars
and years developing the product, called IQOS, which is already
sold in several other countries. It has been waiting for FDA
approval for its U.S. partner Altria Group Inc. to market IQOS to
adult smokers.
IQOS is a hand-held device that heats tobacco but doesn't burn
it, so it doesn't produce smoke when users inhale. It differs from
other smoking alternatives like e-cigarettes that produce vapor
from nicotine-laced liquid.
(END) Dow Jones Newswires
April 30, 2019 14:45 ET (18:45 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
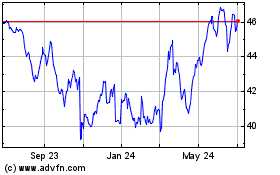
Altria (NYSE:MO)
Historical Stock Chart
From Mar 2024 to Apr 2024
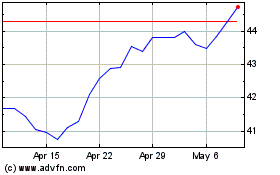
Altria (NYSE:MO)
Historical Stock Chart
From Apr 2023 to Apr 2024