NEW YORK, Oct. 24, 2018 /PRNewswire/ -- Neurotrope Inc.
(NASDAQ: NTRP), a clinical-stage biopharmaceutical company
developing novel therapies for neurodegenerative diseases,
including Alzheimer's disease, today announced the presentation of
updated data from the Phase 2, multidose, exploratory trial
evaluating Bryostatin-1 as a treatment of cognitive deficits in
moderate to severe Alzheimer's disease ("AD"). The data were
presented in a poster, entitled "Evidence of Sustained Low Dose
Bryostatin Efficacy for Treatment of Alzheimer's Disease:
Consistency of Multiple Evaluation Analyses" at the 11th Edition of
Clinical Trials on Alzheimer's Disease ("CTAD") being held
in Barcelona, Spain from
October 24th - 27th, 2018.
"We are encouraged by the findings of this exploratory trial as
we believe they are suggestive of the primary mechanisms of action
of bryostatin, synaptogenesis and anti-apoptosis, or the generation
of new, mature synaptic connections and the prevention of neuronal
death," stated Dr. Daniel Alkon,
President and Chief Scientific Officer of Neurotrope. "These
results show a potential reversal of disease progression occurring
in patients with moderate to severe AD treated with
Bryostatin-1. Notably, this improvement persisted even 30
days after completion of all drug administration."
This study was designed to assess the safety and efficacy of
Bryostatin-1 as a treatment of cognitive deficits in patients with
moderate to severe AD (n=147). Patients enrolled in the study
were allowed to continue on background therapy, including
cholinergic and/or antiglutamatergic treatment. The primary
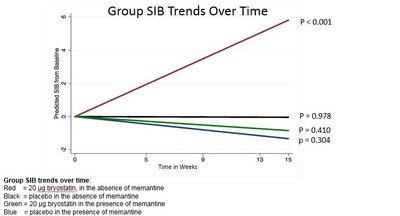
efficacy analysis was change in Severe Impairment Battery (SIB)
scores. For patients in the 20 µg Bryostatin-1 dose group (n=49,
with 38 completers), the dose group being explored in an ongoing
confirmatory Phase 2 study, SIB scores were consistently greater
than baseline, indicating improvement in cognitive function. The
magnitude of improvement was greater in a pre-specified exploratory
analysis of patients not on memantine therapy (n=16).
Memantine is an antiglutamatergic treatment whose mechanism of
action may interfere with that of bryostatin-1. Analyses of
patients in this group showed evidence of sustained SIB improvement
over baseline compared to placebo patients not on memantine (>
6.30 points, 8.4 points when compared to all placebo). Individual
patient trends over time revealed that 15 out of 16 patients (94%)
in the 20μg Bryostatin-1, non-memantine group showed improvement in
SIB by the end of the trial. No increase in SIB measures over time
for the placebo patients or for patients on baseline memantine
therapy was found.
A model for repeated measures (MMRM) was used in the trend
analysis to provide consistency with the analysis of the whole
patient sample. Based on the statistical trend analysis, the 20 μg
Bryostatin-1 in the absence of memantine group shows a significant
positive SIB trend (e.g. SIB improving over time). The
treatment-by-time interaction, indicating a difference in treatment
effect by arm, was highly significant (p < 0.001). These data
indicate that benefit for the patients in the memantine-free group
increase with dosing over time, including throughout the 15 week
protocol.
Neurotrope is evaluating the 20 µg dose of Bryostatin-1 in
patients not on memantine in a confirmatory Phase 2 trial,
initiated in July, 2018, and evaluating 100 moderate-to-severe AD
patients. Data from this study are expected during the second
half of 2019.
The poster presentation information and poster presentation link
are listed below.
Title: Evidence of Sustained Low Dose Bryostatin Efficacy for
Treatment of
Alzheimer's Disease: Consistency of Multiple
Evaluation Analyses
Abstract #: LBP14
Presentation Theme: Clinical trials: Results
Date/Time: Wednesday, October
24th, 2018: 1:00 – 4:00
PM
Location: Palau de Catalunya
Conference Center
About Neurotrope
Neurotrope is at the forefront of developing a new approach to
combating AD and other neurodegenerative diseases. The
Company's world-class science offers the potential to realize a
paradigm shift to overcome one of today's most challenging clinical
problems — finding a way to slow or even prevent the progression of
AD.
In addition to the Company's Phase 2 trial of Bryostatin-1 in
advanced AD, Neurotrope has also conducted preclinical studies of
Bryostatin-1 as a potential treatment for rare diseases and brain
injury, including Fragile X syndrome, multiple sclerosis, stroke,
Niemann-Pick Type C disease, Rett syndrome, and traumatic brain
injury. The FDA has granted Orphan Drug Designation to Neurotrope
for Bryostatin-1 as a treatment for Fragile X. Bryostatin-1
has already undergone testing in more than 1,500 people in cancer
studies, thus creating a large safety data base that will further
inform clinical trial designs.
Please visit www.neurotrope.com for further
information.
Forward-Looking Statements
Any statements contained in this press release that do not
describe historical facts may constitute forward-looking
statements. These forward-looking statements include statements
regarding the Phase 2 study and further studies, and continued
development of use of Bryostatin-1 for Alzheimer's dementia and
other cognitive diseases. Such forward-looking statements are
subject to risks and uncertainties and other influences, many of
which the Company has no control over. There can be no assurance
that the clinical program for Bryostatin-1 will be successful in
demonstrating safety and/or efficacy that we will not encounter
problems or delays in clinical development, or that Bryostatin-1
will ever receive regulatory approval or be successfully
commercialized. Actual results and the timing of certain events and
circumstances may differ materially from those described by the
forward-looking statements as a result of these risks and
uncertainties. Additional factors that may influence or cause
actual results to differ materially from expected or desired
results may include, without limitation, the Company's inability to
obtain adequate financing, the significant length of time
associated with drug development and related insufficient cash
flows and resulting illiquidity, the Company's patent portfolio,
the Company's inability to expand the Company's business,
significant government regulation of pharmaceuticals and the
healthcare industry, lack of product diversification, availability
of the Company's raw materials, existing or increased
competition, stock volatility and illiquidity, and the
Company's failure to implement the Company's business plans or
strategies. These and other factors are identified and described in
more detail in the Company's filings with the SEC, including the
Company's Annual Report on Form 10-K for the year
ended December 31, 2017, and on Form 10-Q for the quarter
ended June 30, 2018. The Company
does not undertake to update these forward-looking statements.
Contact information:
Investors and Media
Sam Martin and Ryan Baker
Argot Partners
212-600-1902
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/neurotrope-presents-updated-phase-2-data-of-bryostatin-1-in-moderate-to-severe-alzheimers-disease-at-the-11th-edition-of-ctad-300736940.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/neurotrope-presents-updated-phase-2-data-of-bryostatin-1-in-moderate-to-severe-alzheimers-disease-at-the-11th-edition-of-ctad-300736940.html
SOURCE Neurotrope Inc.