LabCorp’s William Hanlon Appointed to Governing Committee of the National Evaluation System for health Technology Coordinat...
July 18 2017 - 8:45AM
Business Wire
LabCorp® (NYSE: LH), a leading global life sciences company,
announced today that the National Evaluation System for health
Technology Coordinating Center (NESTcc) has appointed William
(Bill) Hanlon, Ph.D., chief development officer and head of Global
Regulatory Affairs for its Covance Drug Development business, to
serve on its Governing Committee. The Medical Device Innovation
Consortium (MDIC), a public-private partnership created with the
objective of advancing the regulatory science of medical devices
for patient benefit, established NESTcc in 2016 with a grant from
the U.S. Food and Drug Administration (FDA). The FDA has stated its
interest in generating better evidence for medical device
evaluation and regulatory decision-making.
This Smart News Release features multimedia.
View the full release here:
http://www.businesswire.com/news/home/20170718005298/en/
Bill Hanlon, Ph.D., chief development
officer and head of Global Regulatory Affairs, Covance (Photo:
Business Wire)
“Real-world evidence generated from post-market evaluation of
devices used in clinical practice will provide important
information to support the development of new devices and new uses
for currently marketed devices,” said Hanlon. “The sources of these
data are advancing quickly – medical devices, point-of-care
devices, wearables, apps – creating the promise of rich and precise
insights into how patients benefit from these devices as well as
respond to new therapies. It is an honor and singular opportunity
to serve on the NESTcc Governing Committee, which is bringing
together critical stakeholders at this pivotal moment in
healthcare. Together, our goal is to increase the responsible use
of real-world evidence with an inclusive, patient-centered
approach.”
Hanlon is the American Clinical Laboratory Association nominee
to the Governing Committee. He has been an active contributor to
the development of innovative new medicines for almost 30 years,
holding positions of increasing scientific leadership and
responsibility. For the last 15 years, Hanlon has focused on early
and late-stage clinical development as a regulatory affairs expert.
He earned his Bachelor of Science degree in biochemistry from
Rutgers University and his Doctor of Philosophy in biochemistry and
cell biology, jointly conferred by the University of Medicine and
Dentistry of New Jersey and Rutgers University Graduate School.
“Dr. Hanlon’s expertise in regulatory affairs and clinical
development is an important addition to the NEST Coordinating
Center’s Governing Committee,” said NESTcc Executive Director
Rachael Fleurence, Ph.D. “I look forward to working closely with
Dr. Hanlon, who will ensure the perspective of clinical
laboratories is appropriately represented in NEST’s
activities.”
NESTcc’s mission is to support the timely and reliable
development of real-world evidence associated with medical devices
throughout the total product life cycle. Real-world evidence should
be generated in the course of clinical or home care, and will
comply with robust methodological standards. With support from the
Coordinating Center’s executive director and staff, the Governing
Committee will recruit Expert Advisory Working Groups, develop
NESTcc bylaws and undertake the development of shared resources
through specific demonstration projects.
About LabCorp
LabCorp (NYSE: LH), an S&P 500 company, is a leading global
life sciences company that is deeply integrated in guiding patient
care, providing comprehensive clinical laboratory and end-to-end
drug development services. With a mission to improve health and
improve lives, LabCorp delivers world-class diagnostic solutions,
brings innovative medicines to patients faster and uses technology
to improve the delivery of care. LabCorp reported net revenues of
nearly $9.5 billion for 2016 through the contributions of 52,000
employees in approximately 60 countries. To learn more about
LabCorp, visit www.labcorp.com, and to learn more about Covance
Drug Development, visit www.covance.com.
This press release contains forward-looking statements including
with respect to estimated 2017 guidance and the impact of various
factors on operating and financial results. Each of the
forward-looking statements is subject to change based on various
important factors, including without limitation, competitive
actions in the marketplace, and adverse actions of governmental and
other third-party payers. Actual results could differ materially
from those suggested by these forward-looking statements. The
Company has no obligation to provide any updates to these
forward-looking statements even if its expectations change. Further
information on potential factors that could affect operating and
financial results is included in the Company’s Form 10-K for the
year ended December 31, 2016, and subsequent Forms 10-Q, including
in each case under the heading risk factors, and in the Company’s
other filings with the SEC. The information in this press release
should be read in conjunction with a review of the Company’s
filings with the SEC including the information in the Company’s
Form 10-K for the year ended December 31, 2016, and subsequent
Forms 10-Q, under the heading MANAGEMENT’S DISCUSSION AND ANALYSIS
OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170718005298/en/
LabCorpMediaPattie Kushner, +1
336-436-8263Media@labcorp.comorCovance MediaSue Maynard, +1
336-436-8263Media@labcorp.comorInvestorsScott Frommer, +1
336-436-5076Investor@labcorp.com
Laboratory Corporation o... (NYSE:LH)
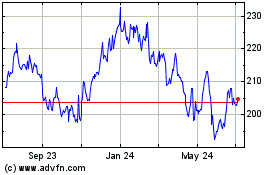
Historical Stock Chart
From Mar 2024 to Apr 2024
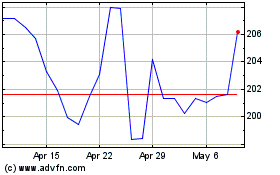
Laboratory Corporation o... (NYSE:LH)
Historical Stock Chart
From Apr 2023 to Apr 2024