Dr. Reddy's Laboratories Announces USFDA Approval for the Launch of Doxorubicin Hydrochloride Liposome Injection in the U.S. ...
May 17 2017 - 4:53AM
Business Wire
Dr. Reddy’s Laboratories Ltd. (BSE: 500124, NSE: DRREDDY, NYSE:
RDY) announced today that it has received approval from the U.S.
Food and Drug Administration (USFDA) to launch Doxorubicin
Hydrochloride Liposome Injection, a therapeutic equivalent generic
version of Doxil® (doxorubicin hydrochloride liposome injection),
for intravenous use, in the United States market. USFDA approval is
an outcome of extensive collaboration with the company’s partner,
Natco Pharma Ltd. (NSE: NATCOPHARM; BSE: 524816), on R&D and
manufacturing capabilities.
“This approval represents the first of its kind for Dr. Reddy’s
in the complex depot injectables arena,” explains Alok Sonig,
Executive Vice President and Head of the North America Generics
business at Dr. Reddy’s Laboratories. “It is a testament to our
commitment to bring affordable generic medicines to market for
patients. The approval further validates our capabilities
to successfully develop and manufacture complex liposomal
formulations. We are preparing for a commercial launch soon.”
“We are pleased with our partnership with Dr. Reddy’s
Laboratories. This approval would not have been possible without
their guidance and support,” says Rajeev Nannapaneni, Vice Chairman
and Chief Executive Officer, Natco Pharma.
The Doxil® brand and generic had U.S. sales of approximately
$196 million MAT for the most recent twelve months ending in March
2017 according to IMS Health*.
Dr. Reddy’s Doxorubicin Hydrochloride Liposome Injection is a
sterile, translucent, red liposomal dispersion in 10-mL or 30-mL
glass, single-dose vials. Each 10-mL vial contains 20 mg
doxorubicin hydrochloride at a concentration of 2 mg/mL. Each 30-mL
vial contains 50 mg doxorubicin hydrochloride at a concentration of
2 mg/mL. The following individually cartoned vials are
available:
mg invial
fillvolume
vial size
20 mgvial
10-mL 10-mL
50 mgvial
25-mL 30-mL
WARNING: CARDIOMYOPATHY and INFUSION
RELATED REACTIONS
See full prescribing information for
complete boxed warning.
- Myocardial damage may lead to congestive heart failure and
may occur as the total cumulative dose of doxorubicin HCl
approaches 550 mg/m2. The risk of cardiomyopathy may
be increased at lower cumulative doses with mediastinal
irradiation.
- Acute infusion-related reactions occurred in 11% of patients
with solid tumors. Serious, life-threatening, and fatal infusion
reactions have been reported. Medications/emergency equipment to
treat such reactions should be available for immediate
use.
DOXIL® is a registered trademark of ALZA Corporation.
*IMS National Sales Perspective: Retail and Non-Retail MAT March
2017RDY-0517-159
About Dr. Reddy’s: Dr. Reddy’s Laboratories Ltd. (BSE:
500124, NSE: DRREDDY, NYSE: RDY) is an integrated pharmaceutical
company, committed to providing affordable and innovative medicines
for healthier lives. Through its three businesses - Pharmaceutical
Services & Active Ingredients, Global Generics and Proprietary
Products – Dr. Reddy’s offers a portfolio of products and services
including APIs, custom pharmaceutical services, generics,
biosimilars and differentiated formulations. Our major therapeutic
areas of focus are gastrointestinal, cardiovascular, diabetology,
oncology, pain management and dermatology. Dr. Reddy’s operates in
markets across the globe. Our major markets include – USA, India,
Russia & CIS countries, and Europe. For more information, log
on to: www.drreddys.com
Disclaimer: This press release may include statements of
future expectations and other forward-looking statements that are
based on the management’s current views and assumptions and involve
known or unknown risks and uncertainties that could cause actual
results, performance or events to differ materially from those
expressed or implied in such statements. In addition to statements
which are forward-looking by reason of context, the words "may,"
"will," "should," "expects," "plans," "intends," "anticipates,"
"believes," "estimates," "predicts," "potential," or "continue" and
similar expressions identify forward-looking statements. Actual
results, performance or events may differ materially from those in
such statements due to without limitation, (i) general economic
conditions such as performance of financial markets, credit
defaults, currency exchange rates, interest rates, persistency
levels and frequency/severity of insured loss events (ii) mortality
and morbidity levels and trends, (iii) changing levels of
competition and general competitive factors, (iv) changes in laws
and regulations and in the policies of central banks and/or
governments, (v) the impact of acquisitions or reorganization,
including related integration issues.
The company assumes no obligation to update any information
contained herein.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170517005482/en/
Dr. Reddy’s Laboratories Ltd.INVESTOR RELATIONSSAUNAK
SAVLA, +91-40-49002135saunaks@drreddys.comorMEDIA
RELATIONSCALVIN PRINTER, +91-40-
49002121calvinprinter@drreddys.com
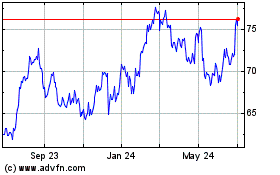
Dr Reddys Laboratories (NYSE:RDY)
Historical Stock Chart
From Mar 2024 to Apr 2024
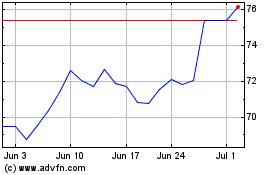
Dr Reddys Laboratories (NYSE:RDY)
Historical Stock Chart
From Apr 2023 to Apr 2024