Avita Medical to Present at Needham Healthcare Conference
March 23 2017 - 8:52PM
Business Wire
Avita Medical Limited (ASX:AVH; OTCQX:AVMXY), a regenerative
medicine company focused on the treatment of wounds and skin
defects, today announced that Adam Kelliher, Avita’s Chief
Executive Officer will present at the 16th Annual Needham
Healthcare Conference on 4 April 2017. The presentation will take
place at 9:20am ET at The Westin Grand Central in New York
City.
Mr. Kelliher’s presentation will update audiences on Avita’s
commercial, regulatory, scientific and clinical research
developments, providing recent data and outcomes across the range
of indications for which the Company’s portfolio of products
(ReCell®, ReGenerCell™ and ReNovaCell™) are in use within various
markets. Mr. Kelliher will also discuss Avita’s contract award from
the Biomedical Advanced Research and Development Authority (BARDA)
to deploy ReCell® as a medical countermeasure for US mass casualty
preparedness. A review of the FDA approval pathway and progress
will also be provided.
Presentation details:
Date:
Tuesday, 4 April, 2017
Time:
9:20am ET
Location:
The Westin Grand Central in New York
ABOUT RECELL® AND RES™
ReCell® is Avita Medical’s unique proprietary technology that
enables a clinician to rapidly create, at point of care in
approximately 30 minutes, Regenerative Epithelial Suspension (RES™)
using a small sample of the patient’s skin. RES™ is an autologous
suspension comprising the cells and wound healing factors necessary
to regenerate natural, healthy skin. RES™ has a broad range of
applications and can be used to restart healing in unresponsive
wounds, to repair burns using less donor skin, yet with improved
functional and aesthetic outcomes, and to restore pigmentation and
improve cosmesis of damaged skin.
ABOUT AVITA MEDICAL LIMITED
Avita Medical develops and distributes regenerative products for
the treatment of a broad range of wounds, scars and skin defects.
Avita’s patented and proprietary collection and application
technology provides innovative treatment solutions derived from a
patient’s own skin. The Company’s lead product, ReCell®, is used in
the treatment of a wide variety of burns, plastic, reconstructive
and cosmetic procedures. ReCell® is patented, CE-marked for Europe,
TGA-registered in Australia, and CFDA-cleared in China. In the
United States, ReCell® is an investigational device limited by
federal law to investigational use, and a pivotal U.S. approval
trial is underway. To learn more, visit www.avitamedical.com.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170323006340/en/
Avita Medical LtdAdam KelliherChief Executive Officer+44
020 8947 9804akelliher@avitamedical.comorTim RooneyChief Financial
Officer+ 1 (661)
367-9170trooney@avitamedical.comorAustraliaMonsoon
CommunicationsSarah Kemter+61 (0)3 9620 3333Mobile: +61 (0)407
162 530sarahk@monsoon.com.auorUSAWestwicke
PartnersJamar Ismail+1 (415)
513-1282jamar.ismail@westwicke.com
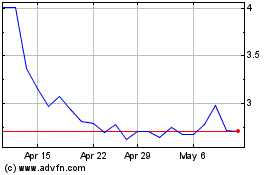
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Mar 2024 to Apr 2024
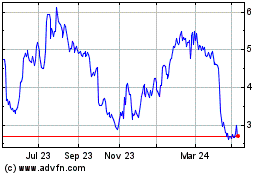
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Apr 2023 to Apr 2024