Anders Hove, MD Joins ARCA biopharma Board of Directors
February 21 2017 - 8:30AM
Business Wire
ARCA biopharma, Inc. (Nasdaq: ABIO), a biopharmaceutical company
applying a precision medicine approach to developing
genetically-targeted therapies for cardiovascular diseases, today
announced that Anders Hove, M.D., has joined its Board of
Directors.
This Smart News Release features multimedia.
View the full release here:
http://www.businesswire.com/news/home/20170221005669/en/
Anders Hove, M.D. (Photo: Business
Wire)
“Anders brings decades of experience building and advising
biopharmaceutical companies,” said Dr. Michael R. Bristow,
President and Chief Executive Officer of ARCA. “With his expertise
on both the corporate and investment sides of the industry, Anders’
input and guidance will be valuable to our Board of Directors as we
continue the development of Gencaro and seek to deliver value to
our stockholders.”
Dr. Hove has more than 20 years of experience investing in and
helping build biopharmaceutical companies. He has served on the
Boards of Directors of numerous private and public companies,
currently serving on the Boards of two private biopharmaceutical
companies. In 2011, Dr. Hove was named to Forbes’ Midas List, which
ranked the top venture capitalists in life sciences and technology.
Dr. Hove received a M.Sc. in Biotechnology Engineering from the
Technical University of Denmark, an M.D. from the University of
Copenhagen and an M.B.A. from the Institut Européen
d'Administration des Affaires, or INSEAD.
“I am honored to join the ARCA Board of Directors,” said Dr.
Hove. “This is an exciting time for the organization with the
GENETIC-AF clinical trial interim efficacy analysis expected later
this year. I believe there is a significant opportunity for
developing genetically-targeted cardiovascular therapeutics, for
which ARCA is well-positioned.”
About ARCA biopharma
ARCA biopharma is dedicated to developing genetically-targeted
therapies for cardiovascular diseases through a precision medicine
approach to drug development. The Company's lead product candidate,
GencaroTM (bucindolol hydrochloride), is an investigational,
pharmacologically unique beta-blocker and mild vasodilator being
developed for atrial fibrillation. ARCA has identified common
genetic variations that it believes predict individual patient
response to Gencaro, giving it the potential to be the first
genetically-targeted atrial fibrillation prevention treatment. ARCA
has a collaboration with Medtronic, Inc. for support of the
GENETIC-AF trial. For more information, please visit
www.arcabio.com.
Safe Harbor Statement
This press release contains "forward-looking statements" for
purposes of the safe harbor provided by the Private Securities
Litigation Reform Act of 1995. These statements include, but are
not limited to, statements regarding, the potential timeline for
GENETIC-AF trial activities, the potential for genetic variations
to predict individual patient response to Gencaro, Gencaro’s
potential to treat atrial fibrillation, future treatment options
for patients with atrial fibrillation, the potential for ARCA to
expand its cardiovascular pipeline, the opportunity related to
developing genetically-targeted cardiovascular therapeutics and the
potential for Gencaro to be the first genetically-targeted atrial
fibrillation prevention treatment. Such statements are based on
management's current expectations and involve risks and
uncertainties. Actual results and performance could differ
materially from those projected in the forward-looking statements
as a result of many factors, including, without limitation, the
risks and uncertainties associated with: the Company's financial
resources and whether they will be sufficient to meet the Company's
business objectives and operational requirements; results of
earlier clinical trials may not be confirmed in future trials, the
protection and market exclusivity provided by the Company’s
intellectual property; risks related to the drug discovery and the
regulatory approval process; and, the impact of competitive
products and technological changes. These and other factors are
identified and described in more detail in ARCA’s filings with the
Securities and Exchange Commission, including without limitation
the Company’s annual report on Form 10-K for the year ended
December 31, 2015, and subsequent filings. The Company disclaims
any intent or obligation to update these forward-looking
statements.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170221005669/en/
ARCA biopharma, Inc.Investor & Media Contact:Derek Cole,
720.940.2163derek.cole@arcabio.com
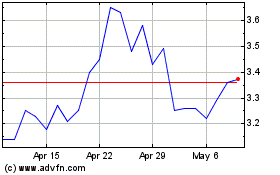
ARCA Biopharma (NASDAQ:ABIO)
Historical Stock Chart
From Mar 2024 to Apr 2024
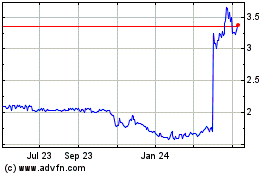
ARCA Biopharma (NASDAQ:ABIO)
Historical Stock Chart
From Apr 2023 to Apr 2024