BETHESDA, Md., Sept. 21, 2016 /PRNewswire/ -- Northwest
Biotherapeutics (NASDAQ: NWBO)("NW Bio"), a U.S.
biotechnology company developing DCVax® personalized immune
therapies for solid tumor cancers, announced that Dr. Marnix Bosch, Chief Technical Officer, today
presented an updated and more detailed analysis of the Phase I
DCVax®-Direct Trial data at the 5th Annual SMi Cancer
Vaccines Conference in London,
England.

DC Vax-Direct is designed to treat inoperable solid
tumors. The Phase I Trial treated patients with multiple
inoperable metastatic tumors who had failed existing treatment
regimens. These patients had failed as many as five or six
different prior types of treatments, and had generally quite poor
life expectancies when they entered the DCVax-Direct Trial.
DCVax-Direct was directly injected, with image guidance, into just
one of the patients' tumors and most patients received just three
treatments. The Trial included 40 patients, with 39
evaluable, and covered more than a dozen diverse types of solid
tumor cancers.
Some highlights of Dr. Bosch's update presentation today
included:
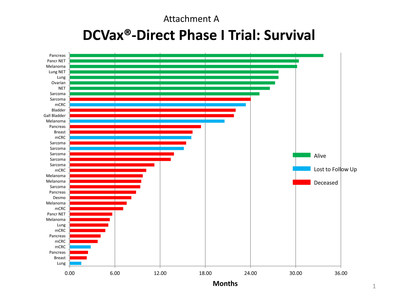
- The top 20% of these patients have so far exceeded 2 years of
survival and are still alive. The longest survivor to date has
reached nearly 3 years. See Attachment A for details.
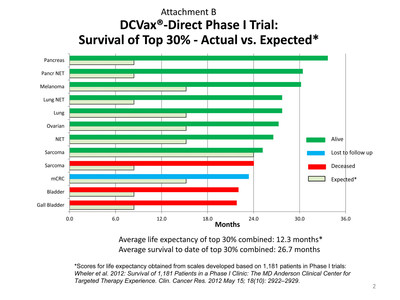
- The top 30% of these patients (including pancreatic, melanoma,
lung, ovarian, sarcoma and other cancers) as a combined group have
an average survival to date of 26.7 months, compared with an
average of expected survival times of 12.3 months.
Individually, these patients have also substantially exceeded
their respective expected survival times. See Attachment B
for details.
- The continuing positive survival results correlate with
underlying mechanisms of action and cellular and immune profiles,
including phenotype analyses, relative production of a wide range
of diverse cytokines by the dendritic cells. Additional
positive observations include T-cell infiltration, and PD-L1
expression.
- 64% of the patients evaluable for PD-L1 checkpoint expression
(14 of 22) showed either de novo or significantly increased
expression of PD-L1 following DCVax-Direct treatment, indicating
potential for combination of DCVax-Direct and checkpoint
inhibitors.
- The diverse cancers covered in this trial are responsible for
approximately 800,000 new cases annually in the US and also at
least 800,000 new cases in Europe.
When these cancers reach the inoperable metastatic stage,
there are no effective treatments available today.
There was no webcast of the presentation, but Dr. Bosch's slides
will be available on the NW Bio website starting today.
About Northwest Biotherapeutics
Northwest Biotherapeutics is a biotechnology company focused on
developing personalized immunotherapy products designed to treat
cancers more effectively than current treatments, without
toxicities of the kind associated with chemotherapies, and on a
cost-effective basis, in both the United
States and Europe. The Company has a broad platform
technology for DCVax dendritic cell-based vaccines. The
Company's lead program is a 348-patient Phase III trial in newly
diagnosed Glioblastoma multiforme (GBM), which is on a partial
clinical hold in regard to new screening of patients. GBM is
the most aggressive and lethal form of brain cancer, and is an
"orphan disease." The Company is under way with a 60-patient
Phase I/II trial with DCVax-Direct for all types of inoperable
solid tumors cancers. It has completed enrollment in the
Phase I portion of the trial. The Company previously conducted
a Phase I/II trial with DCVax-L for metastatic ovarian cancer
together with the University of
Pennsylvania. The Company previously received
clearance from the FDA for a 612-patient Phase III trial in
prostate cancer. In Germany,
the Company has received approval of a 5-year Hospital Exemption
for the treatment of all gliomas (primary brain cancers) outside
the clinical trial.
Disclaimer
Statements made in this news release that are not historical
facts, including statements concerning future treatment of patients
using DCVax and future clinical trials, are forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995. Words such as "expect," "believe,"
"intend," "design," "plan," "continue," "may," "will,"
"anticipate," and similar expressions are intended to identify
forward-looking statements. Actual results may differ
materially from those projected in any forward-looking
statement. Specifically, there are a number of important
factors that could cause actual results to differ materially from
those anticipated, such as risks and uncertainties related to the
actions and decisions of Nasdaq, the Company's ongoing ability to
raise additional capital, risks related to the Company's ability to
enroll patients in its clinical trials and complete the trials on a
timely basis, uncertainties about the clinical trials process
including the actions and decisions of the FDA and other
regulators, uncertainties about the timely performance of third
parties, risks related to whether the Company's products will
demonstrate safety and efficacy, risks related to the Company's and
Cognate's abilities to carry out the intended manufacturing and
expansions contemplated in the Cognate Agreements, risks related to
the Company's ability to carry out the Hospital Exemption program
and risks related to possible reimbursement and pricing.
Additional information on these and other factors, including Risk
Factors, which could affect the Company's results, is included in
its Securities and Exchange Commission ("SEC") filings.
Finally, there may be other factors not mentioned above or included
in the Company's SEC filings that may cause actual results to
differ materially from those projected in any forward-looking
statement. You should not place undue reliance on any
forward-looking statements. The Company assumes no obligation
to update any forward-looking statements as a result of new
information, future events or developments, except as required by
securities laws.
Logo -
http://photos.prnewswire.com/prnh/20110329/SF73084LOGO
Photo - http://photos.prnewswire.com/prnh/20160921/410461
Photo - http://photos.prnewswire.com/prnh/20160921/410462
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/nw-bio-presents-updated-data-from-dcvax-direct-phase-i-trial-at-5th-annual-smi-cancer-vaccines-conference-in-london-300331969.html
SOURCE Northwest Biotherapeutics, Inc.