UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A
(RULE
14a-101)
INFORMATION REQUIRED IN PROXY STATEMENT
SCHEDULE 14A INFORMATION
Proxy Statement Pursuant to Section 14(a) of the
Securities Exchange Act of 1934
(Amendment No. )
|
|
|
|
|
|
|
|
|
Filed by the
Registrant
x
Filed by
a Party other than the Registrant
¨
|
|
|
|
Check the appropriate box:
|
|
|
|
|
¨
|
|
Preliminary Proxy Statement
|
|
|
|
|
¨
|
|
Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2))
|
|
|
|
|
x
|
|
Definitive Proxy Statement
|
|
|
|
|
¨
|
|
Definitive Additional Materials
|
|
|
|
|
¨
|
|
Soliciting Material Pursuant to
§240.14a-12
|
|
|
|
|
|
|
STEMCELLS, INC.
|
|
(Name of Registrant as Specified In Its Charter)
|
|
|
|
|
|
(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
|
|
|
|
Payment of Filing Fee (Check the appropriate box):
|
|
|
|
|
x
|
|
No fee required.
|
|
|
|
|
¨
|
|
Fee computed on table below per Exchange Act Rules 14a-6(i)(4) and 0-11.
|
|
|
|
|
|
|
|
(1)
|
|
Title of each class of securities to which transaction applies:
|
|
|
|
|
|
|
|
|
|
(2)
|
|
Aggregate number of securities to which transaction applies:
|
|
|
|
|
|
|
|
|
|
(3)
|
|
Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing
fee is calculated and state how it was determined):
|
|
|
|
|
|
|
|
|
|
(4)
|
|
Proposed maximum aggregate value of transaction:
|
|
|
|
|
|
|
|
|
|
(5)
|
|
Total fee paid:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
¨
|
|
Fee paid previously with preliminary materials.
|
|
|
|
|
¨
|
|
Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement
number, or the Form or Schedule and the date of its filing.
|
|
|
|
|
|
|
|
(1)
|
|
Amount Previously Paid:
|
|
|
|
|
|
|
|
|
|
(2)
|
|
Form, Schedule or Registration Statement No.:
|
|
|
|
|
|
|
|
|
|
(3)
|
|
Filing Party:
|
|
|
|
|
|
|
|
|
|
(4)
|
|
Date Filed:
|
|
|
|
|
|
|
STEMCELLS, INC.
7707 Gateway Blvd.
Newark, California 94560
NOTICE OF ANNUAL MEETING OF STOCKHOLDERS
To be Held on May 5, 2016
To the Stockholders of STEMCELLS, INC.
Notice is hereby given that the Annual
Meeting of Stockholders of StemCells, Inc. (“StemCells” or the “company”) will be held on Thursday, May 5, 2016, at 2 p.m., local time, at 7707 Gateway Boulevard, Newark, California 94560 for the following purposes:
1. to elect the two Class I directors named in the accompanying proxy materials to serve until the 2019 Annual Meeting of
Stockholders;
2. to consider and vote upon a proposal to ratify the selection of Grant Thornton LLP as independent
public accountants for the company for the fiscal year ending December 31, 2016;
3. to approve an amendment to the
company’s certificate of incorporation to effect a reverse stock split of the company’s issued and outstanding common stock, as further described in Proposal Number 3;
4. to approve an amendment to the company’s certificate of incorporation to reduce the number of authorized shares of common stock
from 225 million to 200 million;
5. to approve the sale by the company of up to $16 million in company common stock, in
accordance with a committed equity line purchase agreement, as further described in Proposal Number 4; and
6. to
transact any and all other business that may properly come before the meeting.
The Board of Directors has fixed the close of
business on Friday, March 11, 2016, as the record date for determining those stockholders who are entitled to notice of, and to vote at, the Annual Meeting of Stockholders and any postponements or adjournments thereof. The stock transfer books
will not be closed between the record date and the date of the meeting. Representation of at least a majority of all outstanding shares of common stock of StemCells is required to constitute a quorum. Accordingly, it is important that your shares be
represented at the meeting.
Please read the proxy materials carefully. All stockholders are invited to attend the Annual
Meeting. Your vote is important, and we appreciate your cooperation in considering and acting on the matters presented.
|
|
|
By Order of the Board of Directors,
|
|
|
|

|
|
Kenneth B. Stratton, J.D.
|
|
Secretary
|
April 8, 2016
Newark, California
PROXY STATEMENT
ANNUAL MEETING OF STOCKHOLDERS
OF
STEMCELLS, INC.
The accompanying proxy is solicited on behalf of the Board of Directors of StemCells, Inc. (the “company”) for use at its
annual meeting of stockholders (the “Annual Meeting”) to be held on Thursday, May 5, 2016, at 2:00 p.m., local time, at the company’s headquarters at 7707 Gateway Boulevard, Newark, California 94560. The company will bear the
cost of solicitation of proxies. Directors, officers and employees of the company may solicit proxies by telephone, facsimile or in person for no additional compensation. The company will reimburse banks, brokerage firms, proxy solicitors, and other
custodians, nominees and fiduciaries for reasonable expenses incurred by them in sending proxy materials to the beneficial owners of shares.
The Board of Directors has fixed the close of business on Friday, March 11, 2016, as the record date for determining stockholders
entitled to notice of, and to vote at, the Annual Meeting or at any postponement or adjournment thereof. There were 112,507,589 shares of our common stock, $.01 par value, outstanding on March 11, 2016, each of which is entitled to
one vote for each share on the matters to be voted upon.
Stockholders are being asked to vote on five proposals at the company’s
2016 Annual Meeting. The proposals to be voted on and related recommendations from the Board of Directors are as follows:
Proposal Number 1 — To elect the two director nominees named in this proxy statement to serve as Class I directors on the
Board until our 2019 annual meeting of stockholders or until that person’s successor is duly elected and qualified. The Board of Directors recommends that you vote “FOR” each of the nominees.
Proposal Number 2 — To ratify the appointment of Grant Thornton LLP as the company’s independent registered public
accounting firm for the fiscal year ending December 31, 2016. The Board of Directors recommends that you vote “FOR” this proposal.
Proposal Number 3 — To approve an amendment to the company’s certificate of incorporation, as further described in Proposal Number
3, to effect a reverse stock split of the company’s issued and outstanding common stock and to authorize the Board of Directors to effect this amendment to the certificate of incorporation, within the Board’s discretion, at any time within
three months after the date stockholder approval for the reverse stock split is obtained, with the exact exchange ratio and timing of the reverse stock split (if at all) to be determined at the discretion of the Board of Directors. The Board of
Directors recommends that you vote “FOR” this proposal.
Proposal Number 4 — To approve an amendment to the
company’s certificate of incorporation, as further described in Proposal Number 4, to reduce the number of authorized shares of company common stock from 225 million to 200 million and to authorized the Board of Directors to effect this
amendment to the certificate of incorporation, within the Board’s discretion, at any time within three months after the date stockholder approval for the reduction in authorized capital is obtained. The Board of Directors recommends that you
vote “FOR” this proposal.
Proposal Number 5 — To approve the sale by the company of up to $16 million in company common
stock, in accordance with a committed equity line purchase agreement, as further described in Proposal Number 5, and to authorize the Board of Directors, within its discretion, to enter into this agreement at any time (if at all) within three months
after the date stockholder approval for the equity line is obtained.
1
In the election of directors, which is Proposal Number 1, you may vote “FOR”
both of the nominees or your vote may be “WITHHELD” with respect to one or both of the nominees. For Proposal Number 2, Proposal Number 3, Proposal Number 4, and Proposal Number 5, you may vote “FOR,” vote
“AGAINST” or “ABSTAIN.” If you “ABSTAIN” as to Proposal Number 2 and Proposal Number 5, the abstention will have no effect. If you “ABSTAIN” as to Proposal Number 3 or Proposal Number 4,
the abstention has the same effect as a vote “AGAINST” the proposal.
Shares of our common stock represented by proxies in
the form enclosed that are properly executed and returned to us and not revoked will be voted as specified in the proxy by the stockholder. In the absence of contrary instructions, or in instances where no specifications are made, the shares will be
voted:
(i) FOR the election as directors of the nominees as described herein under “Proposal Number
1 — Election of Directors;”
(ii) FOR ratification of the selection of accountants as described herein
under “Proposal Number 2 — Ratification of Selection of Independent Public Accountants;”
(iii)
FOR the proposal to approve an amendment to the company’s certificate of incorporation, as described herein, to effect a reverse stock split of the company’s authorized, issued and outstanding common stock (the “Reverse Stock
Split”), as described herein under “Proposal Number 3 — Approval of Amendment to the Company’s Certificate of Incorporation to Effect a Reverse Stock Split;”
(iv) FOR the proposal to approve an amendment to the company’s certificate of incorporation, as described herein, to
reduce the number of authorized shares of common stock to 200,000,000, as described herein under “Proposal Number 4 — Approval of Amendment to the Company’s Certificate of Incorporation to Decrease Authorized
Capital;”
(v) FOR the proposal to approve the sale by the company of up to $16 million in company common stock, as
described herein under “Proposal Number 5 — Approval of $16 Million Equity Line Financing;” and
(vi) in the discretion of the named proxies as to any other matter that may properly come before the Annual Meeting.
Any stockholder signing and delivering a proxy may revoke it at any time before it is voted by delivering to the company’s corporate
secretary a written revocation or a duly executed proxy bearing a date later than the date of the proxy being revoked. Any stockholder attending the Annual Meeting in person may revoke his, her or its proxy and vote his, her or its shares at the
Annual Meeting.
How to vote shares at our 2016 Annual Meeting.
This year, company stockholders may cast their vote in any of the following ways:
Vote by Internet.
Any stockholder can vote over the Internet at www.proxyvote.com by following the
instructions on the Notice or proxy card. Internet voting facilities for stockholders of record will be available 24 hours a day and will close at 11:59 p.m. (EDT) on May 4, 2016.
Vote by Mail.
Any stockholder that receives proxy materials by mail can vote by mail by signing, dating
and mailing the enclosed proxy card in the postage-paid envelope provided. If the envelope is missing, such a
2
stockholder can mail the completed proxy card or voting instruction card to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, New York 11717. The completed card must be
received no later than May 4, 2016.
Voting at the Annual Meeting
. All company stockholders are invited to
attend the Annual Meeting in person. Any stockholder that attends the meeting in person may deliver a completed proxy card in person or vote by completing a ballot, which will be available at the meeting. However, each stockholder intending to vote
in person at the Annual Meeting should note that if his, her or its shares are held in the name of a bank, broker or other nominee, such stockholder must obtain a legal proxy, executed in his, her or its favor, from the holder of record to be able
to vote at the Annual Meeting. Stockholders should allow enough time prior to the Annual Meeting to obtain this proxy from the holder of record, if needed.
The shares voted electronically or represented by the proxy cards received, properly marked, dated, signed and not revoked, will be voted at
the Annual Meeting.
QUORUM, REQUIRED VOTES AND METHOD OF TABULATION
Consistent with Delaware law and the company’s amended and restated by-laws, a majority of the votes entitled to be cast on a particular
matter, present in person or represented by proxy, constitutes a quorum as to such matter. The company will appoint one or more election inspectors for the meeting to count votes cast by proxy or in person at the Annual Meeting.
If you hold shares beneficially in street name and do not provide your broker or nominee with voting instructions, your shares may constitute
“broker non-votes.” Generally, broker non-votes occur on a matter when a broker is not permitted to vote on that matter without instructions from the beneficial owner and instructions have not been given. This year if you hold shares
beneficially in street name and do not vote your shares, your broker or nominee can vote your shares at its discretion on Proposal Number 2, Proposal Number 3 and Proposal Number 4. In tabulating the voting result for any proposal for
which the required vote is based on the number of shares present, shares that constitute broker non-votes are not considered entitled to vote on that proposal. Thus, broker non-votes will not affect the outcome of Proposal Number 1 or
Proposal Number 5 provided a quorum is established.
What vote is required to approve each item?
Election of directors by stockholders, which is Proposal Number 1, will be determined by a plurality of the votes cast by the
stockholders entitled to vote at the election that are either present in person or represented by proxy.
For
Proposal Number 2 and Proposal Number 5, the affirmative “FOR” vote is required by the holders of a majority of the shares present at the Annual Meeting in person or by proxy and voting. Abstentions will have no effect on
the outcome of this proposal.
For Proposal Number 3 and Proposal Number 4, the affirmative “FOR” vote is required by
the holders of a majority of the shares outstanding as of the record date for the 2016 Annual Meeting. For this reason, any abstentions would be the equivalent of a vote against the proposal.
Management does not know of any matters to be presented at this year’s Annual Meeting other than those set forth in this proxy statement
and in the notice accompanying this proxy statement. Stockholders will have no appraisal rights under Delaware law with respect to any of the matters expected to be voted on at the Annual
3
Meeting. If other matters should properly come before the meeting, the proxy holders will vote such matters in their discretion. Any stockholder has the right to revoke his, her or its proxy at
any time until it is voted.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The following table shows the number of shares of our common stock beneficially owned, as of March 1, 2016, by (i) each of our
directors, (ii) each of our named executive officers, (iii) all of our current directors and executive officers as a group, and (iv) all those known by us to be to a beneficial owner of more than 5% of the company’s common stock.
In general, “beneficial ownership” refers to shares that an individual or entity has the power to vote or dispose of, and any rights to acquire common stock that are currently exercisable or will become exercisable within 60 days of
March 1, 2016. Unless otherwise indicated, we believe that each person named below, based on information furnished by such owner, holds sole investment and voting power (or shared voting power with affiliated entities) with respect to such
shares, subject to community property laws where applicable. We calculated percentage ownership in accordance with the rules of the SEC. The percentage of common stock beneficially owned is based on 112,507,589 shares outstanding as of
March 1, 2016. In addition, shares issuable pursuant to options, restricted stock units or other convertible securities that may be acquired within 60 days of March 1, 2016 are deemed to be issued and outstanding and have been treated
as outstanding in calculating and determining the beneficial ownership and percentage ownership of those persons possessing those securities, but not for any other persons.
|
|
|
|
|
|
|
|
|
|
|
Name and Address of Beneficial Owner*
|
|
Amount and
Nature of
Beneficial
Ownership
|
|
|
Percentage of
Class Beneficially
Owned
|
|
|
Directors and Named Executive Officers
|
|
|
|
|
|
|
|
|
|
Eric Bjerkholt(1)
|
|
|
131,417
|
|
|
|
*
|
*
|
|
R. Scott Greer(2)
|
|
|
143,841
|
|
|
|
*
|
*
|
|
Ricardo Levy(3)
|
|
|
116,691
|
|
|
|
*
|
*
|
|
Ian Massey(4)
|
|
|
17,059
|
|
|
|
*
|
*
|
|
Greg Schiffman(5)
|
|
|
187,956
|
|
|
|
*
|
*
|
|
John Schwartz(6)
|
|
|
200,463
|
|
|
|
*
|
*
|
|
Ken Stratton(7)
|
|
|
177,883
|
|
|
|
*
|
*
|
|
Alan Trounson(8)
|
|
|
108,253
|
|
|
|
*
|
*
|
|
Irving Weissman(9)
|
|
|
389,340
|
|
|
|
*
|
*
|
|
All current directors and executive officers as a group
|
|
|
1,472,903
|
|
|
|
1.30
|
%
|
|
5% Stockholders
|
|
|
|
|
|
|
|
|
|
Sabby Management, LLC(10)
|
|
|
4,934,100
|
|
|
|
4.39
|
%
|
|
*
|
The address of all directors and executive officers listed in the table is c/o StemCells, Inc., 7707 Gateway Blvd., Newark, California 94560.
|
|
(1)
|
Includes 4,000 shares issuable upon exercise of fully vested stock options. Includes outright ownership of 127,417 shares.
|
|
(2)
|
Includes outright ownership of 143,841 shares.
|
|
(3)
|
Includes 3,000 shares issuable upon exercise of fully vested stock options. Includes outright ownership of 55,438 shares.
|
|
(4)
|
Includes outright ownership of 17,059 shares in Mr. Massey’s 401(k) plan.
|
|
(5)
|
Includes outright ownership of 170,051 shares and 17,905 shares in Mr. Schiffman’s 401(k) plan.
|
4
|
(6)
|
Includes 4,000 shares issuable upon exercise of fully vested stock options. Includes outright ownership of 196,463 shares.
|
|
(7)
|
Includes 15,000 shares issuable upon exercise of 15,000 fully vested stock options. Includes outright ownership of 127,273 shares and 35,610 shares in Mr. Stratton’s 401(k) plan.
|
|
(8)
|
Includes outright ownership of 108,253 shares.
|
|
(9)
|
Includes 3,000 shares issuable upon exercise of fully vested stock options. Includes outright ownership of 363,869 shares, 22,471 shares held in trust 10,508 of which Dr. Weissman disclaims beneficial ownership.
|
|
(10)
|
Represents (i) Sabby Healthcare Master Fund, Ltd. and Sabby Volatility Master Fund, Ltd. beneficially own 3,634,100 and 1,300,000 shares of our common stock, respectively, representing approximately 3.34% and 1.19% of
our outstanding common stock, respectively, and (ii) Sabby Management, LLC and Hal Mintz each beneficially own 4,934,100 shares of our common Stock, representing approximately 4.53% of our outstanding common stock. Sabby Management, LLC and Hal
Mintz do not directly own any shares of our common stock, but each indirectly owns 4,934,100 shares of our common stock. Sabby Management, LLC, a Delaware limited liability company, indirectly owns 4,934,100 shares of our common stock because it
serves as the investment manager of Sabby Healthcare Volatility Master Fund, Ltd. and Sabby Volatility Warrant Master Fund, Ltd., Cayman Islands companies. Mr. Mintz indirectly owns 4,934,100 shares of our common stock in his capacity as manager of
Sabby Management, LLC.
|
Board of Directors
We currently have seven directors serving on our Board of Directors (our “Board”). In January 2016, Martin McGlynn resigned as
the company’s chief executive officer and as a director of the company. Concurrent with this, the Board appointed Ian Massey, D.Phil., as Mr. McGlynn’s successor and appointed Dr. Massey to the Board. The following table shows
the names, ages, principal occupations, and public company board memberships for the last five years of our directors, as of March 1, 2016:
|
|
|
|
|
|
|
|
|
Eric Bjerkholt
|
|
|
56
|
|
|
Eric Bjerkholt was elected to the Board of Directors in March 2004. He is Executive Vice President, Corporate Development and Finance, and Chief Financial Officer of Sunesis Pharmaceuticals, Inc., a biopharmaceutical
company. Mr. Bjerkholt is a member of the board of directors of Ambrx, Inc. and Corium International, Inc.
|
|
|
|
|
|
R. Scott Greer
|
|
|
57
|
|
|
R. Scott Greer was appointed to the Board of Directors in June 2010. He is currently a principal and managing director of Numenor Ventures LLC, which he founded in 2002 to provide funding and strategic advisory services to
early stage enterprises. Mr. Greer currently serves on the boards of Nektar Therapeutics, Inogen, Inc., Sientra, Inc., and Versartis, Inc.
|
|
|
|
|
|
Ricardo Levy, Ph.D.
|
|
|
71
|
|
|
Ricardo Levy, Ph.D. was elected to the Board of Directors in September 2001. He currently serves as chair of the board of directors of NovoDynamics, Inc., a private company focused on advanced image discovery and predictive
analytics.
|
5
|
|
|
|
|
|
|
|
|
Ian Massey, D.Phil.
|
|
|
65
|
|
|
Ian J. Massey, D.Phil., was elected to the Board of Directors in January 2016. He is President and Chief Executive Officer of the company, a position he has held since January 2016.
|
|
|
|
|
|
John Schwartz, Ph.D.
|
|
|
81
|
|
|
John Schwartz, Ph.D., was elected to the Board of Directors in December 1998 and was elected Chairman of the Board at the same time. He is currently President of Quantum Strategies Management Company, a registered investment
advisor.
|
|
|
|
|
|
Alan Trounson, Ph.D.
|
|
|
70
|
|
|
Alan Trounson, Ph.D., was appointed to the Board of Directors in July 2014. He was the President of the California Institute for Regenerative Medicine from January 2008 until May 2014.
|
|
|
|
|
|
Irving Weissman, M.D.
|
|
|
76
|
|
|
Irving Weissman, M.D., was elected to the Board of Directors in September 1997. He is the Virginia and Daniel K. Ludwig Professor of Cancer Research, Professor of Pathology and Professor of Developmental Biology at Stanford
University and the Director of the Institute of Stem Cell Biology and Regenerative Medicine at Stanford.
|
Because we have a classified board, with each of our directors serving a staggered three-year term, only two
of our directors are standing for reelection at our 2016 Annual Meeting. The following table shows the current composition of the three classes of our Board:
Class I Directors (terms scheduled to expire in 2016, but nominated to stand for reelection at our 2016 Annual Meeting):
|
|
|
|
|
|
|
Eric Bjerkholt
|
|
|
|
R. Scott Greer
|
Class II Directors (terms scheduled to expire in 2017):
|
|
|
|
|
|
|
Ricardo Levy, Ph.D.
|
|
|
|
Alan Trounson, Ph.D.
|
|
|
|
Irving Weissman, M.D.
|
Class III Directors (terms scheduled to expire in 2018)
|
|
|
|
|
|
|
Ian Massey, D.Phil.
|
|
|
|
John Schwartz, Ph.D.
|
The independent members of our Board, as determined by the Board of Directors in accordance with the existing
Nasdaq Listing rules, are Messrs. Bjerkholt and Greer and Drs. Levy, Trounson and Schwartz. The Board of Directors held six regular meetings during the fiscal year ended December 31, 2015. Each of our directors attended more than 75%
of the meetings of the Board of Directors and of the committees on which he served, with the exception of Dr. Trounson, who attended only one meeting of the Strategic Transaction Committee in 2015. While we encourage our directors to attend the
company’s annual stockholder meeting, we not have a policy requiring that they do so. Two of our directors attended the company’s 2015 annual stockholder meeting.
For many years, the roles of chairman and chief executive officer at the company have been separated. We believe that this is appropriate
under current circumstances, because it allows management to make the operating
6
decisions necessary to manage the business, while helping to maintain Board independence so that it can provide an effective oversight function. We feel that this has provided an appropriate
balance of operational focus, flexibility and oversight. Our independent directors meet at regularly scheduled executive sessions without members of management.
Board Committees
Presently, the Board
has four standing committees — the Audit Committee, the Compensation and Stock Option Committee (the “Compensation Committee”), the Corporate Governance and Nominating Committee (the “Corporate Governance Committee”),
and the Strategic Transactions Committee — as well as a single-member committee established under the company’s 2006 and 2013 equity incentive plans and the Company’s 2012 Commencement Incentive Plan. All members of the Audit
Committee, the Compensation Committee, and the Corporate Governance Committee are, and are required by the charters of the respective committees to be, independent as determined under Nasdaq Listing rules.
Audit Committee
.
The Audit Committee is composed of Mr. Bjerkholt and
Drs. Schwartz and Levy. The Audit Committee held five meetings during the fiscal year ended December 31, 2015. The primary function of the Audit Committee is to assist our Board in fulfilling its oversight responsibilities. The committee
does this primarily by reviewing our financial reports and other financial information as well as the company’s systems of internal controls regarding finance, accounting, legal compliance, and ethics that management and the Board have
established. The committee also assesses our auditing, accounting and financial processes more generally. The Audit Committee meets at least quarterly, and at such other times as it finds necessary. It recommends to our Board the appointment of a
firm of independent auditors to audit the financial statements of the company and meets with such personnel of the company to review the scope and the results of the annual audit, the amount of audit fees, the company’s internal accounting
controls, the company’s financial statements contained in this proxy statement, and other related matters. Each of the members of the Audit Committee is independent, and the Board has determined that Mr. Bjerkholt is an “audit
committee financial expert,” as defined in SEC rules. The Audit Committee acts pursuant to a written charter which is available through our website at www.stemcellsinc.com.
Compensation Committee
.
The Compensation Committee is composed of Drs. Schwartz and
Levy and Mr. Bjerkholt. The Compensation Committee held three meetings during the fiscal year ended December 31, 2015. The Compensation Committee makes recommendations to our Board and management concerning salaries in general, determines
executive compensation and, except to the extent that such decisions have been delegated to, and made by, the single-member committee, approves incentive compensation for our employees and consultants. The Compensation Committee acts pursuant to a
written charter which is available through our website at www.stemcellsinc.com.
Corporate Governance
Committee
. Since April 2014, the Corporate Governance Committee has been composed of Drs. Levy and Schwartz and Mr. Greer. The Corporate Governance Committee held one meeting in 2015 to discuss a slate
of actual and potential nominees to the Board of Directors. The committee oversees nominations to the Board and considers the experience, ability and character of potential nominees to serve as directors, as well as particular skills or knowledge
that may be desirable in light of the company’s position at any time. From time to time, the committee has engaged the services of a paid search firm to help the committee identify potential nominees to the Board. The company’s Corporate
Governance Committee and Board seek to nominate and appoint candidates to the Board who have significant business experience, technical expertise or personal attributes, or a combination of these, sufficient to suggest, in the Board’s judgment,
that the candidate would have the ability to help direct the affairs of the company and enhance the Board as a whole. The Committee may identify potential candidates through any reliable means available, including recommendations of past or current
members of the Board from their knowledge of the industry and of the company. The Committee also considers past service on the Board or on the board of directors of other publicly traded or
7
technology focused companies. The committee has not adopted a formulaic approach to evaluating potential nominees to the Board; it does not have a formal policy concerning diversity, for example.
Rather, the committee weighs and considers the experience, expertise, intellect, and judgment of potential nominees irrespective of their race, gender, age, religion, or other personal characteristics. The committee often looks for nominees that can
bring new skill sets or diverse business perspectives. Potential candidates recommended by security holders will be considered as provided in the company’s “Policy Regarding Shareholder Candidates for Nomination as a Director,” which
sets forth the procedures and conditions for such recommendations. This policy is available through our website at www.stemcellsinc.com. The Corporate Governance Committee operates pursuant to a written charter, a copy of which is also available
through our website at www.stemcellsinc.com.
The members of the Corporate Governance Committee have approved the nomination of the
Class I directors standing for reelection at our 2016 Annual Meeting.
Strategic Transactions
Committee
. The Strategic Transactions Committee is composed of Messrs. Bjerkholt and Greer and Drs. Levy and Trounson. The Strategic Transactions Committee held five meetings during the fiscal year ended
December 31, 2015. The Strategic Transactions Committee was created at the suggestion of our Chief Executive Officer in March 2009 to provide advice and direction, on an
ad hoc
basis, on a range of strategic initiatives being
considered at the time. The Strategic Transactions Committee does not have a formal charter. However, the Board of Directors has authorized the Strategic Transactions Committee to be available to advise, consult and participate with management, as
requested by the company’s Chief Executive Officer, with respect to the identification, implementation, evaluation, and negotiation of potential strategic corporate transactions. Since June 2010, the Strategic Transactions Committee has
been the Board’s fourth standing committee, and as such it routinely provides recommendations both to management and to the full Board with regard to such matters as the Strategic Transactions Committee may deem advisable.
The following table shows the members of our four standing Board committees:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Director
|
|
Independent
|
|
|
Audit
Committee
|
|
|
Compensation
Committee
|
|
|
Corporate
Governance
Committee
|
|
|
Strategic
Transactions
Committee
|
|
|
Eric Bjerkholt
|
|
|
Yes
|
|
|
|
Chair
|
|
|
|
ü
|
|
|
|
|
|
|
|
ü
|
|
|
R. Scott Greer
|
|
|
Yes
|
|
|
|
|
|
|
|
|
|
|
|
ü
|
|
|
|
Chair
|
|
|
Ricardo Levy, Ph.D.
|
|
|
Yes
|
|
|
|
ü
|
|
|
|
ü
|
|
|
|
Chair
|
|
|
|
ü
|
|
|
Ian Massey, D.Phil.
|
|
|
No
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
John Schwartz, Ph.D.
|
|
|
Yes
|
|
|
|
ü
|
|
|
|
Chair
|
|
|
|
ü
|
|
|
|
|
|
|
Alan Trounson, Ph.D.
|
|
|
Yes
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
ü
|
|
|
Irving Weissman, M.D.
|
|
|
No
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Director Oversight and Qualifications
While management is responsible for the day-to-day management of the risks the company faces, the Board, as a whole and through its
committees, has responsibility for the oversight of risk management. An important part of risk management is not only understanding the risks facing the company and what steps management is taking to manage those risks, but also understanding what
level of risk is appropriate for the company. In support of this oversight function, the Board receives regular reports from our Chief Executive Officer and members of senior management on operational, financial, legal, and regulatory issues and
risks. The Audit Committee additionally is charged under its charter with oversight of financial risk, including the company’s internal controls, and it receives regular reports from management, the company’s internal auditors and the
company’s independent auditors. The chairman of the Board and independent members of the Board work together to provide strong, independent oversight of the company’s management and affairs through its standing committees and, when
necessary, special meetings of directors.
8
We believe each of our directors brings valuable skills, experience, judgment, and perspectives
to our company. The Board took the following qualifications into consideration, among other things, when nominating or appointing our current directors:
|
|
|
|
|
Eric Bjerkholt
|
|
Mr. Bjerkholt is a financial expert and currently serves as the Executive Vice President, Corporate Development and Finance, and Chief Financial Officer of Sunesis Pharmaceuticals, Inc., a biopharmaceutical company. His business
experience spans more than 20 years, during which time he founded a nutraceutical company and worked as an investment banker. Mr. Bjerkholt currently serves on the board of directors of Ambrx, Inc. and Corium International, Inc. We believe Mr.
Bjerkholt’s qualifications to serve on our Board of Directors include his considerable financial and business experience, especially in the life sciences industry. Mr. Bjerkholt has served on our Board for over twelve years.
|
|
|
|
|
R. Scott Greer
|
|
Mr. Greer was appointed to our Board in June 2010. He is a financial expert with over 25 years of experience in the life sciences industry. He was founder, CEO and Chairman of Abgenix, Inc., a biotechnology company he took
public in 1998 and then sold to Amgen in 2006. Mr. Greer currently serves on the boards of Nektar Therapeutics, Inogen, Inc., Sientra, Inc., and Versartis, Inc. We believe Mr. Greer’s qualifications to serve on our Board include his more than
25 years of experience in the life sciences industry.
|
|
|
|
|
Ricardo Levy, Ph.D.
|
|
Dr. Levy was elected to the Board in September 2001. He currently serves as chair of the board of directors of NovoDynamics, Inc., a private company focused on advanced image discovery and predictive analytics, and on the
Advisory Board of Santa Clara University’s Miller Center for Social Entrepreneurship. He is a lecturer at the Stanford University Chemical Engineering Department. We believe his qualifications to serve on our Board of Directors includes his
more than 40 years of business experience. Dr. Levy has served on our Board for over thirteen years.
|
|
|
|
|
Ian Massey, D.Phil.
|
|
Dr. Massey has been our Chief Executive Officer since January 2016. From March 2015 until January 2016, he served as our President and Chief Operating Officer. Before joining the company, Dr. Massey served as
Chief Operating Officer and President of U.S. Operations of Biotie Therapies Corporation. In 2006, Dr. Massey was the co-founder, President and Chief Executive Officer of Synosia Therapeutics. Dr. Massey’s prior experience in the
pharmaceutical industry included over 25 years first with Syntex and then with Roche, where he held positions of increasing responsibility culminating in Sr. Vice President and Head of Research and Preclinical Development of Roche Palo
Alto.
|
|
|
|
|
John Schwartz, Ph.D.
|
|
Dr. Schwartz has over 40 years of business and legal experience, including several years spent in the 1990s as President and Chief Executive Officer of Systemix, Inc., a cell-based therapeutics company which was acquired by
Novartis in 1997. Before joining Systemix as its Senior Vice President and General Counsel in 1993, Dr. Schwartz served as the Vice President and General Counsel of Stanford University. He currently runs a registered investment advisor firm called
Quantum Strategies Management Company. We believe Dr. Schwartz’s qualifications to serve on our Board of Directors include his over 40 years of business and legal experience in our industry. Dr. Schwartz has served on our Board for over
seventeen years.
|
9
|
|
|
|
|
|
|
|
Alan Trounson, Ph.D.
|
|
Dr. Trounson was elected to our Board in July 2014. He most recently served as President of the California Institute for Regenerative Medicine (CIRM) from 2008 until 2014. Dr. Trounson is acting Chief Executive Officer of
the start-up company Cartherics Pty. Ltd. He is also currently a member of the scientific advisory boards of Cord Blood Registry, Cytomatrix Pty Ltd., and Auxogyn, Inc. He was the Director of the Monash Immunology and Stem Cell
Laboratories, located in Melbourne, from 2004 until 2007. From 2003 to 2006, Dr. Trounson was the founder and executive vice-chairman of the Australian Stem Cell Centre. We believe Dr. Trounson’s qualifications to serve on our Board
include his more than 35 years of experience in the life sciences industry.
|
|
|
|
|
Irving Weissman, M.D.
|
|
Dr. Weissman has been a leader in the stem cell field for over 25 years. He is a professor at Stanford University and serves as the director of the Stanford Institute for Stem Cell Biology and Regenerative Medicine. He co-founded
Systemix in 1988 and Cellerant Therapeutics, Inc., a hematopoietic stem cell development company, in 2001. He is a member of several scientific advisory boards and national science institutes, including the National Academy of Science, the American
Academy of Arts and Science, and the Institute of Medicine of the National Academy of Sciences. We believe Dr. Weissman’s qualifications to serve on our Board of Directors include the fact that he has been a leader in stem cell research for
over 25 years as well as his substantial business experience in our industry. Dr. Weissman has served on our Board for over eighteen years and serves as the chairman of our Scientific Advisory Board.
|
Stockholders who wish to communicate with our Board of Directors or with a particular director may send a
letter to our corporate secretary at the following address: StemCells, Inc., 7707 Gateway Blvd., Newark, California 94560 (c/o Legal Department). Any communication should clearly specify that it is intended to be made to the entire Board or to
one or more of our directors. Our corporate secretary will review all such correspondence and forward to our Board a summary of all such correspondence and copies of all correspondence that, in the opinion of the secretary, deals with the functions
of the Board or committees thereof or that he otherwise determines requires their attention. The secretary maintains a log of all correspondence received by us that is addressed to members of the Board, and any director may at any time review and
request copies of any such correspondence.
Concerns relating to accounting, internal controls or auditing matters will immediately be
brought to the attention of the chairman of the Audit Committee and handled in accordance with established procedures, which are set out in the Audit Committee’s Policy on Receipt, Retention and Treatment of Complaints Regarding Accounting,
Internal Controls and Auditing Matters. A copy of this policy is available through our website at www.stemcellsinc.com.
Executive Officers, Positions
Held
Throughout 2015, our Chief Executive Officer was Martin McGlynn. However, Mr. McGlynn resigned in
January 2016. His successor, Dr. Ian Massey, became our President and Chief Executive Officer on January 18, 2016. Prior to this, Dr. Massey was our President and Chief Operating Officer.
10
Following are the name, age and other information for our named executive officers, as of
March 1, 2016. All company officers have been elected to serve until their successors are elected and qualified or until their earlier resignation or removal.
|
|
|
|
|
|
|
Ian Massey, D.Phil.
President and Chief Executive Officer
|
|
65
|
|
Ian J. Massey, D.Phil., joined the company in March 2015 as the company’s President and Chief Operating Officer. He was appointed President and Chief Executive Officer of the company and
elected to our Board of Directors in January 2016. Prior to joining the company, Dr. Massey served as Chief Operating Officer and President of U.S. Operations of Biotie Therapies Corporation, a publicly traded Finnish company
focused on neurodegenerative and psychiatric disorders. In 2006, Dr. Massey was the co-founder, President and Chief Executive Officer of Synosia Therapeutics. Dr. Massey’s prior experience in the pharmaceutical industry included over 25
years, first with Syntex and then with Roche, where he held positions of increasing responsibility culminating in Sr. Vice President and Head of Research and Preclinical Development of Roche Palo Alto.
|
|
|
|
|
|
Greg Schiffman
Chief Financial Officer and Executive Vice President of Finance
|
|
58
|
|
Greg Schiffman joined the company in January 2014 as Chief Financial Officer and Executive Vice President of Finance, with responsibility for corporate financial management, financial reporting and controls, investor
relations, corporate communications, and information technology. Prior to joining StemCells, Mr. Schiffman served as Executive Vice President and Chief Financial Officer of Dendreon Corporation. From 2006 to 2009, Mr. Schiffman served as a director
of VNUS Technologies, Inc. In November 2014, Dendreon Corporation filed for Chapter 11 bankruptcy protection.
|
|
|
|
|
|
Ken Stratton, J.D.
General Counsel
|
|
47
|
|
Ken Stratton, J.D., joined the company in February 2007 as General Counsel, with responsibility for corporate compliance and legal affairs. In March 2008, he assumed responsibility for the Human Resources function.
Prior to joining StemCells, Mr. Stratton served as Deputy General Counsel for Threshold Pharmaceuticals and as Senior Legal Counsel for Medtronic, Inc.’s Vascular business unit.
|
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), requires our executive officers,
directors, and persons who own more than 10% of a registered class of our equity securities, to file with the SEC reports of ownership of our securities and changes in reported ownership. Executive officers, directors and greater than 10% beneficial
owners are required by SEC rules to furnish us with copies of all Section 16(a) reports they file. Based solely on a review of the copies of such forms furnished to us, or written representations from the reporting persons that no Form 5
was required, we believe that, during the fiscal year ended December 31, 2015, all Section 16(a) filing requirements applicable to our officers, directors and greater than 10% beneficial owners have been met.
11
Code of Business Conduct and Ethics
We have adopted a Code of Ethics and Conduct that applies to all of our directors, officers, employees, and consultants. A copy of our code of
ethics is posted on our website at www.stemcellsinc.com. We intend to disclose any substantive amendment or waivers to this code on our website. There were no substantive amendments or waivers to this code in 2015.
EXECUTIVE COMPENSATION
Compensation Committee
Report
The Compensation Committee has reviewed and discussed with management the foregoing Compensation Discussion and Analysis, as
required by Item 402(b) of Regulation S-K. Based on this review and discussion, the Compensation Committee has recommended to our Board of Directors that the Compensation Discussion and Analysis be included in this proxy statement and our
Annual Report on Form 10-K for the fiscal year ending December 31, 2015.
Compensation Discussion and Analysis
We structure our compensation programs to attract and retain talented employees and reward them for helping us achieve our short-term and
long-term goals. We intend for our compensation programs to be equitable and competitive when measured against those offered by companies against whom we compete for high-level scientific and executive personnel. We also intend for them to link pay
to both company and individual performance.
In seeking to accomplish these objectives, we follow a compensation strategy designed,
ultimately, to reward increasing stockholder value. However, because achievement of our principle mission — the research, development and commercialization of stem cell therapeutics — is a long, expensive and challenging process,
we often set individual compensation by using surrogate endpoints to gauge employee contributions towards building sustained stockholder value, such as:
|
|
•
|
|
the achievement of stated corporate goals adopted from time to time by the Board;
|
|
|
•
|
|
the leadership an executive officer has shown in inspiring and marshaling excellent performances in his or her direct reports;
|
|
|
•
|
|
the anticipation, identification and successful disposition of issues and problems that, if not addressed timely and effectively, might have a deleterious effect on the company; and
|
|
|
•
|
|
the speed and effectiveness with which an executive officer discovers, assesses and, where appropriate, pursues promising opportunities for the company.
|
Compensation elements.
We, like most biotechnology companies, use a combination of base salary, bonuses
and equity awards to compensate our employees, including our executive officers. As a small company — we have approximately 55 employees in total and only three executive officers — we feel that having so few people in each
job classification and level makes it inefficient to establish a formulaic allocation of total compensation among its various elements; we rely, instead, on our experience and judgment.
In exercising this judgment, we periodically collect and review information (i) from third party market reports such as the
Radford
Biotechnology Survey
— Executive Report
; and (ii) from the proxy statements of other similar biotechnology companies, especially those operating in the San Francisco Bay Area, as well as those pursuing
cell-
12
based therapeutics.
1
In the case of the executive officers who report directly to our chief executive officer, we also carefully consider
the recommendations of our chief executive officer when setting compensation. We integrate all of this information with our evaluation of the individual performance of each of our executive officers.
While we believe our officers and other employees are outstanding, we realize that the company is not yet profitable and that it is still in a
relatively early stage of development. We therefore generally prefer to target our compensation practices so that our employees’ base salaries, bonuses, equity compensation, and benefits all fall close to the 50th percentile paid by
comparable companies for similar positions. Actual compensation may fall slightly above or below these targets, however, because of any number of factors such as general economic conditions, market competition for specific jobs, personal
performance, and the need for internal equities within the company. For example, from time to time, we have paid many of our employees, including some of our executive officers, at below the 50th percentile because of poor economic conditions
generally or periods of difficult fundraising markets for biotechnology companies. At the same time, however, we have paid many of our employees, including some of our executive officers, at above the 50th percentile because of highly
competitive demand for workers with their unique skill sets.
Interaction of compensation
elements.
The basic compensation elements — base salary, bonuses and equity awards — are, as noted, standard in our industry. Though not set independently of one other, we use each element as a
portion of total compensation because we believe we would not otherwise be competitive and because we feel that together they are the proper components of a balanced compensation package:
|
|
•
|
|
base salary is compensation for current efforts;
|
|
|
•
|
|
bonuses, whether in cash or equity, are paid at the Board’s discretion typically for achievements in meeting or exceeding corporate goals; and
|
|
|
•
|
|
equity awards are inducements to remain with the company and to build future value.
|
On
occasion, we have considered our employee compensation programs, including our executive compensation programs, and the effect they may have on company risk. We have concluded that our employee compensation programs are simple and straight-forward
and consistent with those of similarly situated research and development companies. In determining that our compensation policies and practices do not present risks that are likely to have a material adverse effect on our business, our directors
have, from time to time, discussed with management the various pay practices used to compensate our employees at both the executive and non-executive levels. These inquiries have included discussions about our three primary components of
compensation, namely base compensation, cash bonuses and equity incentive compensation.
Our Board of Directors has also periodically
considered how bonus awards are determined and calculated by the company, noting that all bonuses are awarded entirely at the discretion of our Board after taking into consideration the progress of our company’s programs. Based on its review,
our Board has concluded that our bonus program properly aligns compensation with our overall goals, all of which are designed to have a positive impact on our business.
In addition, our Board has periodically examined our equity compensation practices, noting that we typically grant customary equity awards
that vest over many years after the date of grant. We believe discretionary compensation that vests over multiple years does not encourage short-term or high-risk
1
In June 2013, for example, we considered the following companies as being close comparables for purposes of evaluating executive compensation: Aastrom Biosciences, Inc.; Advanced Cell
Technology, Inc.; Athersys, Inc.; BioCryst Pharmaceuticals, Inc.; BioTime, Inc.; Cel-Sci Corporation; Cytokinetics, Incorporated; Cytori Therapeutics, Inc.; Geron Corporation; Medicinova, Inc.; Neostem, Inc.; Neuralstem, Inc.; Nova Bay
Pharmaceuticals, Inc.; Osiris Therapeutics, Inc.; and Sangamo Biosciences, Inc.
13
opportunistic behavior and instead aligns our employees’ interests with the long-term interests of our stockholders by encouraging activities intended to build long-term value for the
company.
For these reasons, we have concluded that our employee compensation programs are designed with the appropriate balance of risk
and reward in relation to our company’s overall business strategy and do not incentivize executives or other employees to take unnecessary or excessive risks. As a result, we believe that risks arising from our employee compensation policies
and practices are not reasonably likely to have a material adverse effect on the company.
Other compensation elements and
benefits.
We offer all employees various health and welfare benefit plans. Our executive officers may participate in these on the same terms as other employees. We do not have a pension plan nor do we use
non-qualified deferred compensation.
2
We offer our employees (again, including executive officers on the same terms as others) a 401(k) defined contribution plan, and match employee
contributions on a 1:2 basis (
i.e.
, $1 contribution by the company for every $2 contribution made by the employee) up to a maximum of 3% of the employee’s salary, subject to legal limitations. At this time, our 401(k) match is made in
the form of shares of common stock in the company.
Compensation of Named Executive Officers
Base salary compensation; target bonuses.
We consider base salary to be a critical component of our
executive officers’ overall compensation packages. We intend the salaries of our executive officers to reflect their actual responsibilities and job scope. We also endeavor to set base compensation levels so that their salaries are competitive
with salaries paid by comparable companies to employees with similar experience, taking into account the cost of living in the San Francisco Bay Area. Accordingly, we have made occasional adjustments to the salaries of certain employees to
address perceived below market anomalies, address specific retention concerns or reward special contributions made to the company.
In
addition to base salary, each employee of the company, including each of our named executive officers, is given a personal target bonus (calculated as a percentage of base salary), based upon factors such as seniority, job title and the existing
targets of co-workers with comparable job responsibilities within the company. Bonuses at the company are discretionary and awarded by the Board in its sole discretion. But when bonuses are awarded, we use the personal target of each employee to
calculate his or her bonus amount.
With these various principles in mind, we recently took the following actions with respect to the base
compensation and bonus targets of certain named executive officers: (i) effective June 17, 2013, we increased Mr. McGlynn’s base salary to $570,000 from $550,000 in recognition of contributions made on behalf of the company and market
factors; (ii) effective June 17, 2013, we increased Mr. Stratton’s base salary to $320,000 from $300,000 in recognition of contributions made on behalf of the company and market factors; (iii) effective January 18, 2016, we
increased Dr. Massey’s base salary from $380,000 to $520,000 upon his promotion to Chief Executive Officer in recognition of increased job responsibilities, and (iv) effective January 25, 2016, we increased Mr. Stratton’s base
salary to $340,000 from $320,000 in recognition of contributions made on behalf of the company and market factors. We made no changes to executive officer compensation in 2014 or 2015.
Bonus compensation.
We view periodic bonuses, whether paid in cash or equity, as an important element of
compensation for several reasons. Bonuses help align individual employee efforts with overall corporate strategies and objectives. Bonuses also help us manage salary expense, while still allowing us to reward successes. By using discretionary
bonuses as part of the compensation mix, we have greater flexibility in managing the timing and amounts of compensation. Accordingly, each year we estimate for planning purposes an aggregate “bonus pool,” which is calculated by using the
base salaries of all our employees and their respective
2
Accordingly, we omit tables showing pension benefits and non-qualified deferred
compensation.
14
target bonuses, and which assumes the Board will elect to award each employee 100% of his or her personal target bonus amount for the year. However, as explained below, the actual bonus award for
any particular year is entirely within the Board’s discretion.
From July 2013 through 2015, we did not change the base
compensation or target bonus of any of our existing executive officers. However, Mr. Schiffman, our Chief Financial Officer, joined the company on January 1, 2014 and Dr. Massey joined the company on March 23, 2015, as our
President and Chief Operating Officer. In January 2016, Dr. Massey became our President and Chief Executive Officer and the company increased his base salary and target bonus at that time in recognition of his new roles and responsibilities.
Accordingly, the base compensation and target bonus information for our current executive officers can be summarized as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Year Ended 12/31/14
Base Compensation/
Target Bonus
|
|
|
Year Ended 12/31/15
Base Compensation/
Target Bonus
|
|
|
Current Base
Compensation/
Target Bonus
|
|
|
President and Chief Executive Officer*
|
|
|
—
|
|
|
$
|
380,000/40
|
%
|
|
$
|
520,000/50
|
%
|
|
Chief Financial Officer
|
|
|
—
|
|
|
$
|
450,000/50
|
%
|
|
$
|
450,000/50
|
%
|
|
General Counsel
|
|
$
|
320,000/40
|
%
|
|
$
|
320,000/40
|
%
|
|
$
|
340,000/40
|
%
|
|
*
|
Dr. Massey was the company’s President and Chief Operating Officer from March 2015 until January 2016. He has served as the company’s President and Chief Executive Officer since January 18,
2016.
|
In practice, over the past few years, we have awarded bonuses on an annual basis after considering, among other
things, the company’s accomplishments against stated corporate goals adopted by the Board, the company’s financial position, the status of its development programs, clinical progress and corporate development activities, and general
economic factors. This has necessarily involved a subjective assessment by the Compensation Committee of corporate performance and market conditions each year.
The process of establishing our corporate goals over the past few years has been a lengthy one. For each fiscal year, our executive officers
have presented the Compensation Committee of the Board with approximately five to ten proposed corporate goals, each often consisting of multiple sub-parts. Management has usually presented its recommended corporate goals to the Compensation
Committee concurrent with our proposed corporate budgets for the following fiscal year. Goals have been designed to be challenging, so that one would not expect consistent achievement of all of them. Typically these goals have included some
preclinical and clinical goals for our HuCNS-SC cell-based platform technology, financing and corporate development goals, and goals related to advancement in cell manufacturing practices. While all these goals have been considered important, and we
have used a cross-functional and balanced approach to setting them, we have typically prioritized our goals by assigning relative weightings to each of them, with all of them together adding up to 100%.
After receiving management’s recommended goals, members of the Compensation Committee typically review them with our executive officers
and oftentimes provide suggestions for additional goals or changes to the recommended goals. After our executive officers and directors have completed this iterative process, which has often taken several weeks, the Compensation Committee adopts
revised corporate goals consistent with the foregoing principles and recommends the updated corporate goals to the full Board for consideration and approval.
Thereafter, during each fiscal year, our executive officers have used the Board-approved corporate goals as a management tool, for example to
coordinate activities, motivate personnel and help prioritize the use of company resources. The executive officers have sometimes referred back to the corporate goals when providing business updates to the Board, similar to management’s
reference back to an approved annual budget.
15
Recently, at the end of each fiscal year or shortly afterwards, our chief executive officer (our
“CEO”) has presented the Compensation Committee with his assessments of corporate performance against the Board-approved corporate goals, together with a summary of any important factors that weighed in his assessments, which he has
provided as context.
Because our corporate goals have not been formulaic or quantitative in nature (we have not had a corporate goal tied
to specific stock price, revenues or expenses, for example), our CEO’s assessments have been largely qualitative in nature. Along with these assessments, our CEO has provided a percentage score for each goal reflecting the degree to which each
goal was or was not, in his judgment, achieved during the year.
The Compensation Committee has usually considered these percentage scores
as well as our CEO’s commentary about corporate performance and more general assessments of the state of our business when determining whether to award employees a company-wide corporate bonus in any given year, and if so how much of the
available bonus pool to award. However, the Compensation Committee members have used their own judgment to determine the size of any bonus award, if any. In any given year, the Board may decide in its judgment to award more than 100% of the bonus
pool for the year. The Board may also decide to award less than 100% of the bonus pool, even if all of the corporate goals have been achieved, if it decides doing so would be in the best interests of the company. While the Compensation Committee and
the Board as a whole use the corporate goals as a measure of success, the amount of any bonus grant, as well as how and when it will be paid, is completely within the Board’s sole discretion.
With these various principles in mind, in early 2016 and as part of its annual year-end review of performance, the Compensation Committee
(with input from our CEO and other Board members) considered, among other things, significant company performance accomplishments in 2015, the company’s successes measured against its 2015 corporate goals, the degree of difficulty in achieving
these goals, as well as other events and circumstances that affected performance. The 2015 goals, as approved by our Board, consisted generally of the following: (i) progress in our CNS Program, including activities aimed at Phase II clinical
testing of our proprietary HuCNS-SC cells in spinal cord injury and age related macular degeneration; (ii) progress toward the implementation of certain quality systems; (iii) timely publication of clinical data; (iii) successful
completion of certain product development and GMP manufacturing activities; (iv) partnering and corporate development activities; and (v) successful fundraising efforts.
Highlights of the 2015 accomplishments taken into account by the Compensation Committee in determining the overall company performance
included:
|
|
•
|
|
In April 2015, we completed transplanting the six patients comprising the first cohort of our Phase II Pathway Study. The first cohort is an open-label dose escalation arm to determine the cell dose to be used
for the second cohort of the study.
|
|
|
•
|
|
In April 2015, we raised gross proceeds of approximately $25 million through a public offering of 35,715,000 units.
|
|
|
•
|
|
In May 2015, we presented a summary of the safety and preliminary efficacy data from our Phase I/II study investigating our proprietary HuCNS-SC human neural stem cells as a treatment for chronic thoracic spinal
cord injury. The analysis of the study demonstrated that the surgical transplantation technique and cell dose were safe and well tolerated by all patients. In addition to safety, analysis of the twelve-month data revealed sustained improvements in
sensory function that emerged consistently around three months after transplantation and persisted until the end of the study. Two patients progressed during the study from the most severe classification, AIS A, to the lesser degree of injury grade,
AIS B.
|
16
|
|
•
|
|
In June 2015, we commenced enrollment of the second cohort in our Phase II Pathway Study in spinal cord injury.
|
|
|
•
|
|
In June 2015, we presented a summary of the safety and preliminary efficacy data from our Phase I/II clinical trial in dry age related macular degeneration (AMD). The fifteen patient, open-label, Phase I/II trial
was designed to evaluate the safety and preliminary efficacy of sub-retinal HuCNS-SC cell transplantation in geographic atrophy, the most advanced form of dry AMD.
|
|
|
•
|
|
In July 2015, we transplanted our first subject in our Radiant Study. This Phase II randomized, controlled proof-of-concept study was designed to evaluate both the safety and efficacy of our proprietary HuCNS-SC
cells for the treatment of dry AMD.
|
|
|
•
|
|
In November 2015, we announced that the six-month interim results for the first cohort in our ongoing Phase II Pathway Study in spinal cord injury showed motor improvements in both strength and function. Four of
the six patients showed improvement in the spinal level of injury as defined by the ISNCSCI (International Standards for Neurological Classification of Spinal Cord Injury) assessment of at least one level.
|
|
|
•
|
|
In December 2015, we initiated a strategic realignment plan to fully focus our resources on our proprietary HuCNS-SC cells for the treatment of chronic spinal cord injury.
|
Following this review, the Compensation Committee awarded a discretionary bonus equal to 80% of the available bonus pool, based upon the
committee members’ assessments of market conditions, corporate risks, our market comparables, and the company’s performance in 2015 measured against its 2015 corporate goals, including the successes highlighted above, among other things.
The bonuses were calculated using each employee’s annual base salary as of January 1, 2015. However, in recognition of the company’s cash position in January 2016, the Compensation Committee elected to defer the payment of half
the awarded bonus pool until the completion of the company’s next financing. Furthermore, the Compensation Committee concurred with management’s recommendation to defer 100% of the bonus payout for certain employees of the Company,
including all of the company’s executive officers, until the next successful financing by the company.
Accordingly, in
January 2016, the company awarded, but did not pay, Dr. Massey a 2015 bonus in the amount of $94,615, because on January 1, 2015 his base salary and target bonus were, respectively, $380,000 and 40%, and because he joined the company in
March 2015, so his bonus award was prorated for time worked. The company also awarded, but did not pay, Mr. Schiffman a 2015 bonus in the amount of $180,000, because on January 1, 2015 his base salary and target bonus were,
respectively, $450,000 and 50%. The company also awarded, but did not pay, Mr. Stratton a 2015 bonus in the amount of $102,400, because on January 1, 2015 his base salary and target bonus were, respectively, $320,000 and 40%. Lastly, as
part of his severance agreement with the company, Mr. Mc Glynn was not eligible to receive a 2015 bonus.
Since then, because we
successfully completed an equity financing in March 2016, our Compensation Committee released the second half of the 2015 bonus payment to all our employees, including our executive officers. Our Chief Executive Officer and Chief Financial Officer
elected to receive their 2015 bonuses as common stock equity awards under our 2013 Equity Incentive Plan. The Compensation Committee granted these awards in March 2016.
Equity Compensation
— general practices.
We believe that equity compensation
awards are an important component of our overall compensation policy because equity compensation can provide strong inducement to remain with the company and to build future stockholder value. In order to achieve these objectives, we believe that
equity compensation awards need to be structured to provide both meaningful value and a meaningful
17
opportunity to realize that value. Accordingly, from time to time, we have considered several forms of equity compensation awards, including stock options, stock appreciation rights, restricted
stock, and restricted stock units, because each of these have certain advantages and disadvantages relative to the others with respect to how they might reward effort and success and how they might help us retain high contributors. Generally
speaking, over the years, we have used stock options and restricted stock units (RSUs) as the most common equity compensation instruments. We feel each of these forms of equity has unique and important features for employee retention and for
incentivizing the executive officers to build a profitable and sustainable business.
We have frequently granted either stock option
awards or RSUs to newly hired employees, effective as of their date of hire, and occasionally to existing employees upon their promotion. Both on-hire awards to non-executive officers and awards upon the promotion of current employees are usually
made by either our CEO, acting as the Board’s single-member committee, or by the Compensation Committee. Awards to executive officers are made by either the Compensation Committee or by the full Board. We typically do not grant company-wide
equity awards to employees. Rather, we have granted targeted equity awards to individual employees within the company following a careful review of equity held by each of our employees and the retention value that such awards may provide. In
awarding equity grants to existing employees, we consider their contributions to the company, their roles and responsibilities, their past performance, and market conditions generally for similarly situated employees.
With these various principles in mind, we recently took the following actions with respect to equity compensation.
In December 2012, our Compensation Committee engaged The Croner Company, an executive compensation consultancy firm referred to us by our
outside corporate counsel, to advise the Committee on setting a compensation peer group for the purpose of evaluating the different components of compensation paid to our CEO. During the first half of 2013, our Compensation Committee held six
meetings, many of which were attended by representatives from The Croner Company, to discuss a variety of recommendations made by The Croner Company, including a recommendation that the company establish a new equity incentive plan. The Compensation
Committee also discussed with representatives from both management and The Croner Company a possible peer group for evaluating executive compensation market practices. After considerable discussion and a review of peer information requested by the
Committee, we agreed upon a peer group consisting of fifteen (15) publicly traded companies as being closely comparable to the company because of such business factors as location, nature of operations, industry, and size.
Since these deliberations, and after consideration of our peer group’s compensation practices, we have granted a number of equity awards
to our executive officers and to other key employees. In May and June 2013, for example, we awarded a total of 1,120,000 million restricted stock units, each with four-year vesting, to key employees, including an award of 280,000
restricted stock units to Mr. Stratton. Following this, in June 2013, we granted our chief executive officer 700,000 restricted stock units with four-year vesting in recognition of his anticipated central role in achieving the
company’s short- and long-term growth and future programmatic success.
More recently, in mid-2014, our Board began an initiative to
evaluate our equity compensation practices against an updated list of company comparables. From May to December 2014, the Compensation Committee met three times to discuss various aspects of this initiative. In May 2014, the Committee
consulted Radford, an Aon Hewitt company, about an updated list of peer companies for use by the Committee to evaluate executive compensation at the Company. By August 2014, after considerable discussion and a review of peer information
requested by the Compensation Committee, we agreed upon a peer group consisting of seventeen publicly traded companies as being closely
18
comparable to the company because of such business factors as location, nature of operations, industry, and size.
3
With this information, the
Compensation Committee discussed at length the possibility of awarding a significant grant of equity in 2015 to a larger group of key employees, including our executive officers, with the aim of helping to ensure retention of the Company’s
leadership team and key contributors as the Company conducts its groundbreaking Phase II studies in cervical spinal cord injury and age related macular degeneration, with both of these studies planned to complete by the end of 2017. Given the
importance of both the timely and successful completion of these studies, the Compensation Committee agreed, after further discussions with our CEO, that a substantive equity award to the leadership team and key contributors, especially one with an
element of vesting tied to the clinical studies, would be in the best interest of our stockholders.
Following this determination, the
Committee tasked the company’s CEO to provide possible milestones relating to the planned Phase II clinical studies, which he thought to be appropriate, measurable, and achievable if the company conducts the studies in a thoughtful and
expeditious manner. With these proposed milestones in hand, our Compensation Committee gave special consideration to the comparative benefits of equity awards which vest over time and are therefore believed to have strong retention value to the
individual holder (time-based vesting), versus equity awards which only vest upon the achievement of predetermined development milestones and therefore believed to tie executive compensation more closely with programmatic success and building
stockholder value (performance-based vesting). Following considerable discussion and after having reviewed the existing equity awards held by key company employees, including our executive officers, market comparables, as well as the company’s
prospects and goals for the immediate future, the Compensation Committee determined that it would be in the best interest of the company’s stockholders to award equity with both time-based and performance-based vesting.
Subsequently, in December 2014, the Committee approved the grant of restricted stock units to acquire up to approximately six million
shares of common stock with approximately half of the restricted stock units vesting in equal installments on an annual basis over three years and approximately half of the restricted stock units with vesting tied to the timely and successful
conduct and completion of the company’s Phase II clinical studies in spinal cord injury and dry AMD. Our CEO was given express discretion to decide when to grant these awards and, acting in his capacity as the Single Member Committee under our
equity incentive plans, to determine the exact ratio of time-based vesting and performance-based vesting of each employee’s awards other than to our executive officers. With respect to our executive officers, our Compensation Committee
determined that the ratio between restricted stock units with time-based vesting versus performance-based vesting should be closer to 2:1 given that our executive officers typically have responsibility over a wide range of activities at the company,
including financing and business development, prosecuting and protecting our intellectual property, compliance, and managing our operations outside of clinical development. We were also mindful not to overly link equity compensation with clinical
results, especially for those responsible for making a fair assessment of clinical data. Finally, our Board gave our CEO, in his capacity as the Single Member Committee, to set different performance milestones to certain employees within the
company, if he felt that doing so was appropriate given their roles within the company.
3
By August 2014, based in part upon information supplied by Radford at the
Compensation Committee’s request, the company considered the following companies as being close comparables for purposes of evaluating executive compensation: Advanced Cell Technology, Inc.; Athersys, Inc.; BioTime, Inc.; Cel-Sci Corporation;
Cytokinetics, Incorporated; Cytori Therapeutics, Inc.; Dynavax Technologies Corporation; Fate Therapeutics, Inc.; Fibrocell Science Inc.; Five Prime Therapeutics; Geron Corporation; Neostem, Inc.; Neuralstem, Inc.; Osiris Therapeutics, Inc.;
Threshold Pharmaceuticals, Inc.; and Verastem, Inc.
19
With these principles in mind, we awarded our executive officers the following equity awards in
2015:
|
|
|
|
|
|
|
|
|
|
|
|
|
Time-based vesting RSUs(1)
|
|
|
Performance-based vesting RSUs(2)
|
|
|
Martin McGlynn
|
|
|
1,255,500
|
|
|
|
418,500
|
|
|
Ian Massey
|
|
|
—
|
|
|
|
750,000
|
|
|
Greg Schiffman
|
|
|
178,000
|
|
|
|
250,000
|
|
|
Ken Stratton
|
|
|
240,000
|
|
|
|
80,000
|
|
|
(1)
|
Vesting over three years, with one third vesting on each anniversary of the grant.
|
|
(2)
|
Originally, vesting was tied to the timely and successful conduct and completion of the company’s Phase II clinical studies in spinal cord injury and dry AMD, with the opportunity to vest over three years. However,
in January 2016, in recognition of our decision to discontinue enrolling patients into our Radiant Study in dry AMD, the Compensation Committee approved an amendment to certain outstanding equity awards, including those held by our executive
officers, to tie all performance-based vesting identified here to the timely and successful completion of the company’s Phase II Pathway Study.
|
Moreover, we issued restricted stock units to approximately twenty other employees within the company to acquire up to approximately
3 million shares of company common stock, in aggregate, with approximately one third of these awards being time-based vesting and approximately two thirds being performance-based vesting. Of the approximately two million restricted stock
units granted to our non-officers in January 2015, approximately one third are tied to performance milestones unrelated to our clinical programs.
In January 2016, upon his appointment as the company’s President and Chief Executive Officer, we awarded Dr. Massey an additional
restricted stock units to receive up to an additional 1,250,000 shares of company stock upon the achievement of the following performance milestones: (i) 875,000 RSUs will vest upon the timely and successful conduct and completion of the
company’s Phase II clinical study in spinal cord injury, (ii) 250,000 RSUs will vest upon the achievement of a process development milestone, and (iii) 125,000 RSUs will vest upon the achievement of a corporate partnering milestone.
Employment, Severance and Change-in-Control Agreements
Employment agreements of Certain Named Executive Officers.
Mr. McGlynn joined the company as our
president and chief executive officer on January 15, 2001. Under the terms of an employment agreement between Mr. McGlynn and the company, dated January 2, 2001, as amended, Mr. McGlynn received an initial annual base salary of
$275,000 per year, reviewable annually by the Board of Directors, and a bonus, in the Board’s sole discretion, of up to 25% of his base salary. Over time, however, we have increased Mr. McGlynn’s base salary and target bonus so that
they were, respectively, $570,000 and 55% of his base salary, throughout 2015. In his original employment agreement, we also agreed to pay Mr. McGlynn a $50,000 relocation bonus and to reimburse him for relocation expenses, which we did. From
January 2009 through January 2016, we also paid Mr. McGlynn an annual car allowance of $10,000. In January 2016, Mr. McGlynn resigned from the company. As part of his exit, on January 10, 2016, Mr. McGlynn and the
company entered into a separation and consulting agreement, under which Mr. McGlynn received a one-time lump sum payment of $570,000 and accelerated vesting of all his then outstanding equity awards with time-based vesting milestones, as well as
salary continuation for 12 months at his base wage rate of $570,000 per annum.
Dr. Massey joined the company in March 2015 as our
President and Chief Operating Officer. Under the terms of the initial employment agreement between Dr. Massey and the company, dated March 6, 2015, Dr. Massey received an annual base salary of $380,000 per year and a discretionary target bonus
of up to 40% of his base salary. Pursuant to Dr. Massey’s February 2015 employment agreement, we granted him 750,000 restricted stock units
20
under our 2012 Commencement Incentive Plan. This award had performance based vesting tied to the timely and successful conduct and completion of the Phase II clinical studies in spinal cord
injury and dry AMD with the opportunity to vest over three years. In December 2015, however, the company announced the suspension of further patient enrollment in the company’s Phase II study in dry AMD. Therefore, in connection with his
appointment as Chief Executive Officer, the company agreed to an amendment to Dr. Massey’s employment agreement. Under the terms of this January 2016 amendment, in recognition of his new employment responsibilities as the company’s
CEO, Dr. Massey’s annual base salary increased to $520,000 per year and his discretionary target bonus increased to 50%. Also, in the first quarter of 2016 and in connection with his appointment as Chief Executive Officer, the Compensation
Committee awarded Dr. Massey an additional 1,250,000 shares of company stock upon the achievement of the following performance milestones: (i) 875,000 RSUs will vest upon the timely and successful conduct and completion of the
company’s Phase II clinical study in spinal cord injury, (ii) 250,000 RSUs will vest upon the achievement of a process development milestone, and (iii) 125,000 RSUs will vest upon the achievement of a corporate partnering milestone.
Mr. Schiffman joined the company in January 2014 as our Chief Financial Officer and Executive Vice President of Finance. Under the
terms of an employment agreement between Mr. Schiffman and the company, dated November 13, 2013, Mr. Schiffman receives an annual base salary of $450,000 per year and a discretionary target bonus of up to 50% of his base salary.
Pursuant to Mr. Schiffman’s November 2013 employment agreement, we granted him 350,000 restricted stock units under our 2012 Commencement Incentive Plan. This award will vest over four years, with 25% vesting on each of the first four
anniversaries of his employment.
Mr. Stratton joined the company in February 2007 as our general counsel. Under the terms of
his agreement with the company, dated February 2, 2007, Mr. Stratton received an initial annual base salary of $220,000 per year, with a target bonus of up to 20% of his base salary. Over time, however, we have increased
Mr. Stratton’s base salary and target bonus so that they are, respectively, $340,000 and 40% of his base salary. Pursuant to Mr. Stratton’s February 2007 employment agreement, we granted him an option to purchase
15,000 shares of our common stock. This option vested over 48 months, with one-fourth of the shares having vested on the first anniversary of the date on which Mr. Stratton’s employment began and with the remaining shares having
vested at the rate of 1/48th per month on the last day of each month during the ensuing 36 months.
Severance
arrangements.
Each of our executive officers has entered into a severance agreement with the company under which he would receive payments upon termination of his employment by us without cause or consequent to a
change of control.
As part of his separation from the company, Mr. McGlynn and the company entered into a separation and consulting
agreement, dated January 10, 2016, under which Mr. McGlynn received a one-time lump sum payment of $570,000 and accelerated vesting of all his then outstanding equity awards with time-based vesting milestones, as well as salary continuation for
12 months at his base wage rate of $570,000 per annum. No other severance payments are owed.
In the case of Dr. Massey,
pursuant to the terms and conditions of his employment agreement, as amended on January 14, 2016 and upon his becoming the company’s President and Chief Executive Officer, upon either involuntary termination without cause or Dr.
Massey’s resignation for good reason, we would continue to pay his salary and provide benefits for twelve months, at his base salary rate then in effect. In addition, in the event that Dr. Massey becomes disabled and unable to perform the
essential functions of his job, the company will continue to pay his salary for a period of up to six months. In addition, in the even that there is a material diminution in Dr. Massey’s job duties following a corporate change or control or
similar corporate transaction, as defined by his employment agreement, then the company will owe him a lump sum payment, subject to certain qualifications, equal to: (i) a 50% bonus payout, plus (ii) two times his then current yearly base salary,
plus (iii)
21
the projected cost of healthcare continuation for two years, plus (iv) a tax gross up; in this event, any unvested options and any other stock awards held by him would vest upon termination.
In the case of Mr. Schiffman, upon involuntary termination without cause, we would continue to pay his salary and provide benefits for
twelve months, at his base salary rate then in effect. In the event there is a company change of control and either Mr. Schiffman’s employment is involuntarily terminated or he terminates his employment because his job responsibilities
have been materially and adversely impacted, we would continue to pay Mr. Schiffman’s salary and provide benefits for twelve months; in this event, any unvested options and any other stock awards held by him would vest upon termination.
In the case of Mr. Stratton, upon involuntary termination without cause, we would continue to pay his salary and provide benefits
for six months, at his base salary rate then in effect. If the termination were associated with a change of control, we would continue to pay Mr. Stratton’s salary and provide benefits for twelve months; in this event, any unvested options
and any other stock awards held by him would vest upon termination.
If we terminate the employment of any executive officer for cause, or
if the officer resigns without good cause, he would not be entitled to any severance or other benefits.
22
Executive Officer Compensation Tables
The following tables set forth information with respect to the compensation of certain named executive officers for the fiscal years ended
December 31, 2015, 2014 and 2013.
Summary Compensation Table
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name and Principal Position
|
|
Year
|
|
|
Salary
($)(1)
|
|
|
Bonus
($)(2)
|
|
|
Stock
Awards
($)(3)
|
|
|
All Other
Compensation
($)(4)
|
|
|
Total ($)
|
|
|
Martin McGlynn
|
|
|
2015
|
|
|
|
570,000
|
|
|
|
—
|
|
|
|
1,908,360
|
|
|
|
44,362
|
|
|
|
2,522,722
|
|
|
Former Chief Executive Officer
|
|
|
2014
|
|
|
|
570,000
|
|
|
|
219,450
|
|
|
|
—
|
|
|
|
43,334
|
|
|
|
832,784
|
|
|
|
|
|
2013
|
|
|
|
550,000
|
|
|
|
287,375
|
|
|
|
1,239,000
|
|
|
|
43,258
|
|
|
|
2,119,633
|
|
|
|
|
|
|
|
|
|
|
Ian Massey, D.Phil.
|
|
|
2015
|
|
|
|
291,569
|
|
|
|
94,615
|
|
|
|
765,000
|
|
|
|
10,690
|
|
|
|
1,161,874
|
|
|
President and Chief Executive Officer
|
|
|
2014
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
2013
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Gregory Schiffman
|
|
|
2015
|
|
|
|
450,000
|
|
|
|
180,000
|
|
|
|
487,920
|
|
|
|
28,114
|
|
|
|
1,146,034
|
|
|
Chief Financial Officer and EVP of Finance
|
|
|
2014
|
|
|
|
450,000
|
|
|
|
157,500
|
|
|
|
458,500
|
|
|
|
28,110
|
|
|
|
1,094,110
|
|
|
|
|
|
2013
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
Ken Stratton, J.D.
|
|
|
2015
|
|
|
|
320,000
|
|
|
|
102,400
|
|
|
|
364,800
|
|
|
|
33,510
|
|
|
|
820,710
|
|
|
General Counsel
|
|
|
2014
|
|
|
|
320,000
|
|
|
|
89,600
|
|
|
|
—
|
|
|
|
32,580
|
|
|
|
442,180
|
|
|
|
|
|
2013
|
|
|
|
300,000
|
|
|
|
114,000
|
|
|
|
512,400
|
|
|
|
32,652
|
|
|
|
959,052
|
|
|
(1)
|
We pay salaries on a bi-weekly basis. There were 26 pay periods in 2015, 2014 and 2013.
|
|
(2)
|
Each employee’s target bonus is based on his or her salary as of January 1 of the year to which it applies. For 2013, the Board awarded 95% of the target bonus for all company employees. For 2014, the Board
awarded 70% of the target bonus for all company employees. For 2015, the Board awarded 80% of the target bonus for all company employees. For further description of the non-equity incentive plan see the discussion in our “Compensation
Discussion and Analysis” and “Compensation of Named Executive Officers,” above.
|
|
(3)
|
Amounts shown represent the full grant date value of the equity awards granted in each year as computed in accordance with FASB ASC Topic 718. Assumptions used in the calculation of these amounts are included in
Note 9, “Stock-Based Compensation” in our Form 10-K for the period ended December 31, 2015, filed with the SEC on March 14
, 2016.
|
|
(4)
|
The amounts shown in the “All Other Compensation” column for 2014 include the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name and Principal Position
|
|
Employer
Match on
Defined
Contribution
Plans
($)(a)
|
|
|
Transportation
Allowance ($)
|
|
|
Employee
Health and
Welfare
Benefit
Plans ($)(b)
|
|
|
Total
All Other
Compensation
|
|
|
Martin McGlynn
|
|
|
7,950
|
|
|
|
10,000
|
|
|
|
26,412
|
|
|
|
44,362
|
|
|
Former Chief Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ian Massey, D.Phil.
|
|
|
7,950
|
|
|
|
—
|
|
|
|
2,740
|
|
|
|
10,690
|
|
|
President and Chief Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gregory Schiffman
|
|
|
7,950
|
|
|
|
—
|
|
|
|
20,164
|
|
|
|
28,114
|
|
|
Chief Financial Officer and EVP of Finance
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ken Stratton, J.D.
|
|
|
7,950
|
|
|
|
—
|
|
|
|
25,560
|
|
|
|
33,510
|
|
|
General Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
23
|
(a)
|
Under a 401(k) plan, which is open to substantially all of our employees, we make matching contributions in the form of company common stock based on each participant’s voluntary salary deferrals, subject to plan
and legal limits. We match participant contributions on a 1:2 basis up to a maximum of 3% of the employee’s salary. Registered stock is valued and transferred to the employee’s 401(k) account at the end of calendar each quarter.
|
|
(b)
|
We offer all employees various health and welfare benefit plans. Our executive officers may participate in these on the same terms as other employees.
|
24
Outstanding Equity Awards at Fiscal 2015 Year-End
The following tables show equity awards held by certain of our named executive officers as of December 31, 2015:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name
|
|
Option
Awards
Option
Grant
Date
|
|
|
Underlying
Unexercised
Options
(#)
Exercisable(1)
|
|
|
Underlying
Unexercised
Options
(#)
Unexercisable(1)
|
|
|
Option
Exercise
Price
($/sh)(1)
|
|
|
|
|
|
|
|
|
Martin McGlynn
|
|
|
6/25/2010
|
|
|
|
30,000
|
|
|
|
30,000
|
|
|
$
|
10.50
|
|
|
Former Chief Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ian Massey, D.Phil.
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
President and Chief Executive Officer
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gregory Schiffman
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
|
—
|
|
|
Chief Financial Officer and EVP of Finance
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ken Stratton, J.D.
|
|
|
2/28/2007
|
|
|
|
15,000
|
|
|
|
15,000
|
|
|
$
|
26.20
|
|
|
General Counsel
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock Awards
|
|
|
Name
|
|
Date of Award
|
|
|
Number of
Securities
Underlying
Restricted
Stock Units
That Have Not
Vested (2)
#
|
|
|
Market Value of
Securities of
Restricted
Stock Units
That Have Not
Vested (3)
$
|
|
|
Martin McGlynn
|
|
|
6/13/2013
|
|
|
|
350,000
|
|
|
$
|
147,000
|
|
|
Former Chief Executive Officer
|
|
|
1/8/2015
|
|
|
|
1,000,000
|
|
|
$
|
420,000
|
|
|
|
|
|
1/8/2015
|
|
|
|
255,500
|
|
|
$
|
107,310
|
|
|
|
|
|
1/8/2015
|
|
|
|
104,625
|
|
|
$
|
43,943
|
|
|
|
|
|
1/8/2015
|
|
|
|
104,625
|
|
|
$
|
43,943
|
|
|
|
|
|
1/8/2015
|
|
|
|
104,625
|
|
|
$
|
43,943
|
|
|
|
|
|
1/8/2015
|
|
|
|
104,625
|
|
|
$
|
43,943
|
|
|
|
|
|
|
|
Ian Massey, D.Phil.
|
|
|
3/23/2015
|
|
|
|
187,500
|
|
|
$
|
78,750
|
|
|
President and Chief Executive Officer
|
|
|
3/23/2015
|
|
|
|
187,500
|
|
|
$
|
78,750
|
|
|
|
|
|
3/23/2015
|
|
|
|
187,500
|
|
|
$
|
78,750
|
|
|
|
|
|
3/23/2015
|
|
|
|
187,500
|
|
|
$
|
78,750
|
|
|
|
|
|
|
|
Gregory Schiffman
|
|
|
1/1/2014
|
|
|
|
262,500
|
|
|
$
|
110,250
|
|
|
Chief Financial Officer and EVP of Finance
|
|
|
1/8/2015
|
|
|
|
178,000
|
|
|
$
|
74,760
|
|
|
|
|
|
1/8/2015
|
|
|
|
62,500
|
|
|
$
|
26,250
|
|
|
|
|
|
1/8/2015
|
|
|
|
62,500
|
|
|
$
|
26,250
|
|
|
|
|
|
1/8/2015
|
|
|
|
62,500
|
|
|
$
|
26,250
|
|
|
|
|
|
1/8/2015
|
|
|
|
62,500
|
|
|
$
|
26,250
|
|
|
|
|
|
|
|
Ken Stratton, J.D.
|
|
|
1/27/2012
|
|
|
|
32,500
|
|
|
$
|
13,650
|
|
|
General Counsel
|
|
|
5/24/2013
|
|
|
|
140,000
|
|
|
$
|
58,800
|
|
|
|
|
|
1/8/2015
|
|
|
|
240,000
|
|
|
$
|
100,800
|
|
|
|
|
|
1/8/2015
|
|
|
|
20,000
|
|
|
$
|
8,400
|
|
|
|
|
|
1/8/2015
|
|
|
|
20,000
|
|
|
$
|
8,400
|
|
|
|
|
|
1/8/2015
|
|
|
|
20,000
|
|
|
$
|
8,400
|
|
|
|
|
|
1/8/2015
|
|
|
|
20,000
|
|
|
$
|
8,400
|
|
|
(1)
|
Unless otherwise noted, options are granted at the close of market price on the grant date (or on an adjacent market trading day if the Nasdaq Capital Market is closed on the grant date). They vest over a period of four
years as follows: one-fourth of the option vests on the first anniversary of the grant date and 1/48th of the original grant vests each additional month of service.
|
25
|
(2)
|
Restricted stock units granted under our 2006 Equity Incentive Plan, 2013 Equity Incentive Plan and 2012 Commencement Plan. These restricted stock units vest ratably over a three to four-year period on each grant date
anniversary, or upon the achievement of operational milestones.
|
|
(3)
|
Based on the per share closing market price of $0.94 for our common stock on December 31, 2015.
|
Option Exercises and Stock Awards Vested in 2015
The following table contains information about stock options exercised and restricted stock awards vested by each of our named executive
officers during 2015.
Option Exercises and Stock Awards Vested Table
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Option Awards
|
|
|
Stock Awards
|
|
|
Name
|
|
Number of
shares acquired
on exercise
|
|
|
Value realized
on exercise
|
|
|
Number of
shares acquired
on vesting
|
|
|
Value realized
on vesting
|
|
|
Martin McGlynn
|
|
|
—
|
|
|
$
|
—
|
|
|
|
310,333
|
|
|
$
|
267,306
|
|
|
Ian Massey
|
|
|
—
|
|
|
$
|
—
|
|
|
|
—
|
|
|
$
|
—
|
|
|
Gregory Schiffman
|
|
|
—
|
|
|
$
|
—
|
|
|
|
87,500
|
|
|
$
|
90,125
|
|
|
Ken Stratton, J.D.
|
|
|
—
|
|
|
$
|
—
|
|
|
|
102,500
|
|
|
$
|
82,375
|
|
Director Compensation
Cash Compensation.
From December 2009 through 2013, outside directors received quarterly retainers
for Board service in the amount of $6,250 ($12,500 for the chairman of the Board). The chairs of the standing committees received quarterly stipends of either $2,500 (Audit Committee) or $1,250 (Compensation Committee, Corporate Governance Committee
and, since it became a standing committee in June 2010, the Strategic Transactions Committee). Non-employee directors also received $2,000 for each board meeting attended in person or by videoconference and $1,000 for each board meeting
attended by phone, as well as $1,000 for each standing committee meeting attended in person or by videoconference and $500 for each committee meeting attended by phone. Board service fees were typically paid in cash. However, in March 2011, the
Board approved a sub-plan under our 2006 Amended and Restated Equity Incentive Plan, called the Directors’ Fee Plan, which permits directors to elect to receive all or a portion of their board fees in the form of company common stock. We issued
39,950 shares of stock under our Directors’ Fee Plan as consideration for Board service in 2012. We issued 12,745 shares of stock under our Directors’ Fee Plan as consideration for Board service in 2013. We issued no shares of stock under
our Directors’ Fee Plan in 2014 or 2015.
In mid-2013, we asked Dr. Levy, the chairman of our Corporate Governance Committee, to
review board compensation practices at the company. In particular, we wanted to assess whether we were being responsive to market dynamics, as we had not adjusted director compensation for almost four years. To evaluate current market practices,
Dr. Levy assembled a working group consisting of himself, the chairman of our Compensation Committee and our CEO, who worked together with company management to analyze the compensation practices at the fifteen companies previously identified
by The Croner Company as being our closest market
26
comparables.
4
At the working group’s request, the company prepared for the Board’s review a broad survey of director compensation
paid by these market comparables. From this information, we determined that our annual retainers paid for board and committee service fell below the median (i.e., 50th percentile) and in some ways below the 25th percentile of cash compensation
paid by comparable companies to their outside directors, even though our total board cash compensation, including meeting attendance fees, was only slightly below the median paid by our market comparables. Moreover, we determined that the equity
compensation paid to our outside directors was significantly below the market median, with the full value of our annual awards being less than half the median paid by our closest market comparables. In addition to reviewing this market data, the
working group discussed among other things the relative value of continued use of restricted stock units as director compensation, the company’s interest in making director compensation more competitive, as well as the company’s business
prospects and agenda more generally.
Following this, in December 2013, Dr. Levy presented to the Board a series of
recommendations from the working group to change both the cash and equity compensation paid to our outside directors. The Board considered in particular the market data from the comparable companies collected by management, as recommended by The
Croner Company and adopted by the working group. With respect to cash compensation, in recognition of the fact that the company’s director compensation fell below the average compensation paid by our market comparables and to help garner
interest in board service, the Board approved the recommended changes to the cash-based compensation to increase expected cash compensation from $36,000 to $40,000 per year, consisting of both quarterly retainers and regular board attendance fees,
effective April 1, 2014.
Accordingly, our outside directors now receive quarterly retainers for Board service in the amount of
$7,500 ($15,000 for the chairman of the Board). Also, the chairs of our standing committees receive quarterly stipends of $2,500 (Audit Committee), $1,500 (Compensation Committee and Corporate Governance Committee), and $1,250 (Strategic
Transactions Committee). Non-employee directors also receive $2,000 for each board meeting attended in person or by videoconference and $1,000 for each board meeting attended by phone. Members of the standing committees receive attendance fees, as
follows: $2,000 per Audit Committee meeting attended in person ($1,000 for attendance by phone), $1,500 per Compensation Committee or Corporate Governance Committee meeting attended in person ($750 for attendance by phone), and $1,000 per Strategic
Transactions Committee attended in person ($500 for attendance by phone).
Directors are also reimbursed for their expenses in attending
meetings of the Board and meetings of committees of the Board; and have been for many years.
Equity
Compensation
. From January 1, 2012 through 2013, each of the company’s non-employee directors, other than the chairman of the Board, received an annual grant on each anniversary of his or her appointment to the
Board in the form of 10,000 restricted stock units, vesting on the first anniversary of the grant. In addition, the chairman of the Board received an annual grant of 15,000 restricted stock units, vesting on the first anniversary of the grant.
However, in December 2013, following the review of market comparables by the chairs of our Corporate Governance and Compensation
Committees and our CEO described above, the Board replaced the annual grants of 10,000 restricted stock units (15,000 for our chairman) with annual grants, on January 1st of each year for all outside directors, of restricted stock units worth
$60,000, in each case with one-year vesting. Because we had previously made annual equity awards to our outside directors on their board service anniversary dates, the Board
4
As explained above, by June 2013, and with the advice and guidance of The Croner Company, we determined that the following companies are close comparables for purposes of evaluating our
compensation practices: Aastrom Biosciences, Inc.; Advanced Cell Technology, Inc.; Athersys, Inc.; BioCryst Pharmaceuticals, Inc.; BioTime, Inc.; Cel-Sci Corporation; Cytokinetics, Incorporated; Cytori Therapeutics, Inc.; Geron Corporation;
Medicinova, Inc.; Neostem, Inc.; Neuralstem, Inc.; Nova Bay Pharmaceuticals, Inc.; Osiris Therapeutics, Inc.; and Sangamo Biosciences, Inc.
27
agreed to reduce the 2014 awards to the extent each director’s 2013 award already compensated him for Board service through part of 2014. Accordingly, on January 1, 2014, we made the
following grants of restricted stock units: 47,164 restricted stock units to Mr. Bjerkholt; 44,588 restricted stock units to Mr. Greer; 41,438 restricted stock units to Dr. Levy; 44,383 restricted stock units to Dr. Schwartz; and
41,301 restricted stock units to Dr. Weissman. Then, in January 2015, we began awarding each of our non-employee directors restricted stock units worth $60,000 at the beginning of each year as partial compensation for board service.
Accordingly, on January 1, 2015, we awarded each non-employee director 58,253 restricted stock units, which was equal to $60,000 divided by the then current trading price of our stock. On January 1, 2016, we awarded each non-employee
director 146,342 restricted stock units, which was equal to $60,000 divided by the then current trading price of our stock.
28
Director Compensation Table
The following table summarizes cash-based and equity compensation information for our outside directors, including annual Board and committee
retainer fees and meeting attendance fees, for the year ended December 31, 2015:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name
|
|
Fees Earned
or Paid
in Cash
($)
|
|
|
Stock
Awards
($)(1)
|
|
|
All Other
Compensation
($)
|
|
|
Total
($)
|
|
|
Eric Bjerkholt
|
|
|
64,250
|
(2)
|
|
|
60,000
|
(3)
|
|
|
—
|
|
|
|
124,250
|
|
|
R. Scott Greer
|
|
|
46,500
|
(4)
|
|
|
60,000
|
(5)
|
|
|
|
|
|
|
106,500
|
|
|
Ricardo Levy, Ph.D.
|
|
|
59,250
|
(6)
|
|
|
60,000
|
(7)
|
|
|
—
|
|
|
|
119,250
|
|
|
John Schwartz, Ph.D.
|
|
|
84,750
|
(8)
|
|
|
60,000
|
(9)
|
|
|
—
|
|
|
|
144,750
|
|
|
Alan Trounson, Ph.D.
|
|
|
40,500
|
(10)
|
|
|
60,000
|
(11)
|
|
|
|
|
|
|
100,500
|
|
|
Irving Weissman, M.D.
|
|
|
39,000
|
(12)
|
|
|
60,000
|
(13)
|
|
|
50,000
|
(14)
|
|
|
99,000
|
|
|
(1)
|
The amounts shown in this column represent the full grant date fair value of restricted stock unit grants in 2015 as computed in accordance with FASB ASC Topic 718.
|
|
(2)
|
Includes an annual retainer of $30,000, a fee for Mr. Bjerkholt’s role on the Audit Committee of $10,000, and additional fees of $24,250 for Board and committee meetings attended. Also includes $17,000 earned
in 2014 but paid in 2015.
|
|
(3)
|
Mr. Bjerkholt was granted 58,253 restricted stock units on January 1, 2015, vesting on the one year anniversary of the grant. As of December 31, 2015, he had restricted stock units outstanding for the
purchase of 58,253 shares of common stock, in the aggregate.
|
|
(4)
|
Includes an annual retainer of $30,000, a fee for Mr. Greer’s role on the Strategic Transactions Committee of $5,000, and additional fees of $11,500 for Board and committee meetings attended. Also includes
$12,250 earned in 2014 but paid in 2015.
|
|
(5)
|
Mr. Greer was granted 58,253 restricted stock units on January 1, 2015, vesting on the one year anniversary of the grant. As of December 31, 2015, he had restricted stock units outstanding for the
purchase of 58,253 shares of common stock, in the aggregate.
|
|
(6)
|
Includes an annual retainer of $30,000, a fee for Dr. Levy’s role on the Corporate Governance Committee of $6,000, and additional fees of $23,250 for Board and committee meetings attended. Also includes
$16,000 earned in 2014 but paid in 2015.
|
|
(7)
|
Dr. Levy was granted 58,253 restricted stock units on January 1, 2015, vesting on the one year anniversary of the grant. As of December 31, 2015, Dr. Levy had stock options and restricted stock units
outstanding for the purchase of 59,253 shares of common stock, in the aggregate.
|
|
(8)
|
Includes an annual retainer of $60,000, a fee for Dr. Schwartz’s role on the Compensation Committee of $6,000, and additional fees of $18,750 for Board and committee meetings attended. Also includes $22,000
earned in 2014 but paid in 2015.
|
|
(9)
|
Dr. Schwartz was granted 58,253 restricted stock units on January 1, 2015, vesting on the one year anniversary of the grant. As of December 31, 2015, Dr. Schwartz had stock options and restricted
stock units outstanding for the purchase of 59,253 shares of common stock, in the aggregate.
|
|
(10)
|
Includes an annual retainer of $30,000 and additional fees of $10,500 for Board and Committee meetings attended. Also includes $10,500 earned in 2014 but paid in 2015.
|
|
(11)
|
Dr. Trounson was granted 58,253 restricted stock units on January 1, 2015, vesting on the one year anniversary of the grant. As of December 31, 2015, Dr. Trounson had restricted stock units
outstanding for the purchase of 158,253 shares of common stock, in the aggregate.
|
29
|
(12)
|
Includes an annual retainer of $30,000 and additional fees of $9,000 for Board and committee meetings attended. Also includes $9,500 earned in 2014 but paid in 2015.
|
|
(13)
|
Dr. Weissman was granted 58,253 restricted stock units on January 1, 2015, vesting on the one year anniversary of the grant. As of December 31, 2015, Dr. Weissman had stock options and restricted
stock units outstanding for the purchase of 59,253 shares of common stock, in the aggregate.
|
|
(14)
|
Dr. Weissman receives $50,000 per year for his services as a consultant and as the chairman of our Scientific Advisory Board. Since May 2011, Dr. Weissman has received his consultancy fees under this
agreement in the form of company common stock.
|
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Related parties can include any of our directors or executive officers, certain of our stockholders and their immediate family members. Each
year, we prepare and require our directors and executive officers to complete Director and Officer Questionnaires identifying any transactions with us in which the officer or director or their family members have an interest. This helps us identify
potential conflicts of interest. A conflict of interest occurs when an individual’s private interest interferes, or appears to interfere, in any way with the interests of the company as a whole. Our code of ethics requires all directors,
officers and employees who may have a potential or apparent conflict of interest to immediately notify our general counsel, who serves as our compliance officer. In addition, the Corporate Governance Committee of the Board of Directors is
responsible for considering and reporting to the Board any questions of possible conflicts of interest of Board members. Our code of ethics further requires pre-clearance before any employee, officer or director engages in any personal or business
activity that may raise concerns about conflict, potential conflict or apparent conflict of interest. Copies of our code of ethics and the Corporate Governance Committee charter are posted on the corporate governance section of our website at
www.stemcellsinc.com.
In evaluating related party transactions and potential conflicts of interest, our compliance officer and
independent directors apply the same standards of good faith and fiduciary duty they apply to their general responsibilities. They will approve a related party transaction only when, in their good faith judgment, the transaction is in the best
interest of the company.
Dr. Weissman, a member of the Board of Directors, was retained in September 1997 to serve as a
consultant to us. Pursuant to his consulting agreement, Dr. Weissman provides consulting services to us and serves on our Scientific Advisory Board. In return, we pay Dr. Weissman $50,000 per year for his services. We also agreed to
nominate Dr. Weissman for a position on the Board of Directors, and he agreed to serve if elected. Since October 1, 2000, he has been compensated for this service in the same manner and amount as other non-employee members of the Board.
The consulting agreement with Dr. Weissman contains confidentiality, non-competition, and assignment of invention provisions and is for a term of fifteen years, subject to earlier termination by either party. In May 2011,
Dr. Weissman’s consulting agreement was amended by the parties so that the company could choose to pay fees owed to Dr. Weissman under the agreement in the form of company common stock.
PRINCIPAL ACCOUNTING FEES AND SERVICES
Audit and Tax Fees
The Board of
Directors, upon the recommendation of the Audit Committee, has selected the independent accounting firm of Grant Thornton LLP to audit the accounts of the company for the year ending December 31, 2016.
30
The Audit Committee considered the tax compliance services provided by Grant Thornton LLP,
concluded that provision of such services is compatible with maintaining the independence of the independent accountants, and approved the provision by Grant Thornton LLP of tax compliance services with respect to the year ending December 31,
2015.
The Audit Committee received the following information concerning the fees of the independent accountants for the years ended
December 31, 2014 and 2015, has considered whether the provision of these services is compatible with independence of the independent accountants, and concluded that it is:
|
|
|
|
|
|
|
|
|
|
|
|
|
Year Ended
|
|
|
|
|
12/31/15
|
|
|
12/31/14
|
|
|
Audit fees(1)
|
|
$
|
423,623
|
|
|
$
|
429,400
|
|
|
Tax fees
|
|
$
|
43,337
|
|
|
$
|
52,530
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
$
|
466,960
|
|
|
$
|
481,930
|
|
|
(1)
|
Audit fees represents fees for the integrated audit of our annual consolidated financial statements and reviews of the interim consolidated financial statements, and review of audit-related SEC filings; also includes
fees related to issuing comfort letter(s).
|
Audit and tax fees include administrative overhead charges and reimbursement for
out-of-pocket expenses.
Pre-Approval Policies and Procedures
The Audit Committee has adopted policies and procedures for pre-approving all services (audit and non-audit) performed by our independent auditors. In
accordance with such policies and procedures, the Audit Committee is required to pre-approve all audit and non-audit services to be performed by the independent auditors in order to assure that the provision of such services is in accordance with
the rules and regulations of the SEC and does not impair the auditors’ independence. Under the policy, pre-approval is generally provided up to one year and any pre-approval is detailed as to the particular service or category of services and
is subject to a specific budget. In addition, the Audit Committee may pre-approve additional services on a case-by-case basis. During 2015 and 2014, all services performed by our independent auditors were pre-approved.
31
REPORT OF THE AUDIT COMMITTEE
The Audit Committee oversees our accounting and financial reporting processes and the audits of our financial statements on behalf of the
Board, and selects an independent public accounting firm to perform these audits. Management has the primary responsibility for establishing and maintaining adequate internal control over financial reporting, preparing the financial statements, and
establishing and maintaining adequate controls over public reporting. Our independent registered public accounting firm for fiscal 2015, Grant Thornton LLP, had responsibility for conducting an audit of our annual financial statements in accordance
with the standards of the Public Company Accounting Oversight Board (United States) and expressing an opinion on the conformity of those audited financial statements with generally accepted accounting principles.
The Audit Committee oversaw the independent public accounting firm’s qualifications and independence, as well as its performance. The
Audit Committee assisted the Board in overseeing the preparation of the company’s financial statements, the company’s compliance with legal and regulatory requirements, and the performance of the company’s internal audit function. The
Audit Committee met with personnel of the company and Grant Thornton LLP to review the scope and the results of the annual audit, the amount of audit fees, the company’s internal accounting controls, the company’s financial statements
contained in the company’s Annual Report to Stockholders and other related matters.
The Audit Committee has reviewed and discussed
with management the financial statements for fiscal year 2015 audited by Grant Thornton LLP, as well as management’s report on internal control over financial reporting, using the criteria set forth by the Committee of Sponsoring Organizations
of the Treadway Commission (COSO) in Internal Control — Integrated Framework. The Audit Committee has discussed with Grant Thornton LLP various matters related to the financial statements, including those matters required to be discussed
by SAS 114 (The Auditor’s Communication with Those Charged with Governance). The Audit Committee has also discussed with Grant Thornton LLP its report on internal control over financial reporting, has received the written disclosures and the
letter from Grant Thornton LLP required by Public Company Accounting Oversight Board (PCAOB) Ethics and Independence Rule 3526,
Communication with Audit Committees Concerning Independence
(Rule 3526), and has
discussed with Grant Thornton LLP its independence.
Based upon such review and discussions, the Audit Committee recommended to the Board
of Directors, and the Board approved the recommendation, that the audited financial statements be included in the company’s Annual Report on Form 10-K for the fiscal year ending December 31, 2015 for filing with the SEC.
|
|
|
AUDIT COMMITTEE
|
|
|
|
Eric Bjerkholt, Chairman
|
|
Ricardo Levy, Ph.D.
|
|
John Schwartz, Ph.D.
|
32
PROPOSAL NUMBER 1
Election of Directors
The number of directors is currently fixed at seven. Both our restated certificate of incorporation, as amended to date, and our amended and
restated by-laws provide for the classification of the Board of Directors into three classes (Class I, Class II and Class III), as nearly equal in number as possible, with the term of office of one class expiring each year.
Unless otherwise instructed, the enclosed proxy will be voted to elect the nominees named below, who are now Class I directors, as
Class I directors for a term of three years expiring at the 2019 Annual Meeting of Stockholders and until their successors are duly elected and qualified. Both Class I director nominees have been recommended by the Board of Directors
because of their past experience serving on the company’s Board of Directors, the breadth of their business expertise, sound judgment, and demonstrated leadership, among other things. In prior years, the Class II and Class III
directors were nominated for appointment to the Board for similar reasons. Proxies cannot be voted for a greater number of persons than the number of nominees named below. It is expected that the nominees will be able to serve, but if any are unable
to serve, the proxy will be voted for a substitute nominee or nominees designated by the Board of Directors.
The nominees for election as
Class I directors are as follows:
NOMINEES FOR ELECTION AS CLASS I DIRECTORS
|
|
|
|
|
|
|
|
|
Name
|
|
Principal Occupation
|
|
Age*
|
|
Position
|
|
Eric Bjerkholt
|
|
Executive Vice President and CFO, Sunesis Pharmaceuticals, Inc.
|
|
56
|
|
Director
|
|
R. Scott Greer
|
|
Managing Director, Numenor Ventures LLC
|
|
57
|
|
Director
|
|
*
|
Ages are as of March 1, 2016.
|
Eric Bjerkholt
. Eric Bjerkholt was elected to the
Board of Directors of StemCells, Inc. in March 2004. Mr. Bjerkholt chairs our Audit Committee. He is Executive Vice President, Corporate Development and Finance, and Chief Financial Officer of Sunesis Pharmaceuticals, Inc., a small
molecule biopharmaceutical company in South San Francisco, California. Before joining Sunesis, Mr. Bjerkholt served as the Senior Vice President and Chief Financial Officer of IntraBiotics Pharmaceuticals, Inc. Previously, Mr. Bjerkholt co-founded
LifeSpring Nutrition, Inc., a privately held nutraceutical company, and served as its Chief Financial Officer, and later as its President and Chief Executive Officer. From 1990 to 1997, Mr. Bjerkholt was an investment banker at J.P. Morgan &
Co., Inc. Mr. Bjerkholt holds an MBA from Harvard Business School and a Cand.oecon degree in economics and econometrics from the University of Oslo, Norway. He is a member of the board of directors of Ambrx, Inc. and Corium International, Inc.
R. Scott Greer
. R. Scott Greer was elected to the Board of Directors of the Company in June 2010, and was simultaneously appointed
to Chair the Company’s Strategic Transactions Committee. Mr. Greer is currently a principal and managing director of Numenor Ventures LLC, which he founded in 2002 to provide funding and strategic advisory services to early stage enterprises.
He is also an investor and consultant to CEOs and founders of companies in various stages of development. Previously, Mr. Greer was founder of Abgenix, Inc., a biotechnology company that pioneered the development of antibodies as therapeutics, and
served as its Chief Executive Office from its inception in 1996 until 2002, and as its Chairman from 2000 until 2006. Abgenix was
33
acquired by Amgen in 2006 for $2.2 billion. Prior to Abgenix’s formation, Mr. Greer held senior management positions at Cell Genesys, Inc. including Senior Vice President, Corporate
Development and Chief Financial Officer, and held various positions at Genetics Institute (acquired by Wyeth). Mr. Greer serves on the boards of Nektar Therapeutics, Sientra, Inc., Versartis, Inc., and Inogen, Inc. In the past, Mr. Greer served
on several other public and private company boards, including Sirna Therapeutics (acquired by Merck) and Ablexis, companies where he served as Chairman of the Board, Affymax, Anaptys Biosciences, Auspex Pharmaceuticals, Inc., Illumina, Chimeros, and
CV Therapeutics (acquired by Gilead Sciences). Mr. Greer received a B.A. in Economics from Whitman College and an M.B.A. from Harvard University, and was also a Certified Public Accountant.
THE BOARD OF DIRECTORS RECOMMENDS THAT STOCKHOLDERS VOTE
FOR
THE ELECTION OF THE NOMINEES DESCRIBED ABOVE.
34
PROPOSAL NUMBER 2
Ratification of Selection of Independent Public Accountants
The company is asking the stockholders to ratify the selection of Grant Thornton LLP as the company’s independent public accountants for
the fiscal year ending December 31, 2016. The affirmative vote of the holders of a majority of the shares represented and voting at the Annual Meeting will be required to ratify the selection of Grant Thornton LLP.
In the event the stockholders fail to ratify the appointment, the Audit Committee of the Board of Directors will consider it as a
recommendation to select other auditors for the subsequent year, which the Audit Committee would then take under advisement. Even if the selection is ratified, the Audit Committee of the Board at its discretion could decide to terminate the
engagement of Grant Thornton LLP and engage another firm at any time if the Audit Committee determines that such a change would be necessary or desirable in the best interests of the company and its stockholders.
A representative of Grant Thornton LLP is expected to attend the Annual Meeting and is not expected to make a statement, but will be available
to respond to appropriate questions and may make a statement if such representative desires to do so.
THE BOARD OF DIRECTORS RECOMMENDS THAT
STOCKHOLDERS VOTE
FOR
THE PROPOSAL TO RATIFY THE SELECTION OF GRANT THORNTON LLP AS THE COMPANY’S INDEPENDENT PUBLIC ACCOUNTANTS FOR THE FISCAL YEAR ENDING DECEMBER 31, 2016.
35
PROPOSAL NUMBER 3
Approval of Amendment to the Company’s
Certificate of Incorporation to Effect a Reverse Stock Split
General
Our Board of Directors has
unanimously adopted a resolution declaring advisable and recommending to the stockholders for their approval a proposal to amend the company’s restated certificate of incorporation, as amended to date, to effect a reverse stock split of the
company’s issued and outstanding common stock at any whole number ratio between, and inclusive of, one for ten and one for fifteen (the “Reverse Stock Split”). Approval of this Proposal Number 3 would grant our Board the authority,
without further action by the stockholders, to carry out the Reverse Stock Split, at any time within three months after the date stockholder approval for the Reverse Stock Split is obtained from our stockholders, with the exact exchange ratio and
timing of the Reverse Stock Split (if at all) to be determined at our Board’s discretion. Our Board’s decision whether or not (and when) to effect a Reverse Stock Split (and at what whole number ratio to effect the Reverse Stock Split)
will be based on a number of factors, including market conditions, existing and anticipated trading prices for our common stock and the continued listing requirements of the NASDAQ Capital Market.
A sample form of the certificate of amendment relating to this Proposal Number 3, which we would file with the Secretary of State of the State
of Delaware to carry out the Reverse Stock Split, is attached to this proxy statement as Schedule 1 (the “First Amendment”).
As
explained below, we are asking our stockholders to approve this Proposal Number 3 because we believe a Reverse Stock Split would result in a higher price per share for the outstanding shares of our common stock, which should enable us to maintain
our listing on NASDAQ and make our stock more marketable to investors, retail and institutional alike, as investors often have restrictions associated with, or concerns about investing in, “penny” or other low priced stocks. In addition,
as explained below, the Reverse Stock Split, if approved by our stockholders and implemented by our Board, would result in a significant increase in the number of authorized shares of common stock available to us for future issuance to fund our
continued operations and to grow our business.
What to Expect from a Reverse Stock Split
If approved by our stockholders and Board, the Reverse Stock Split would be implemented simultaneously for all of our then-outstanding common
stock (the “Old Shares”) and the exchange ratio would be the same for all of our issued and outstanding shares of common stock. The Reverse Stock Split would affect all of our stockholders uniformly and would not affect any
stockholder’s percentage ownership interests in the company, except to the extent that the Reverse Stock Split results in any of our stockholders owning a fractional share, because fractional shares would be rounded up to the nearest whole
share. Shares of common stock issued pursuant to the Reverse Stock Split (the “New Shares”) would remain fully paid and nonassessable. The Reverse Stock Split would not affect our continuing to be subject to the periodic reporting
requirements of the Securities Exchange Act of 1934, as amended. Upon becoming effective, the Reverse Stock Split would automatically convert outstanding Old Shares into a smaller fraction of New Shares, depending upon which conversion ratio our
Board may select. Outstanding derivative securities exercisable for, or convertible into, our common stock would be proportionally adjusted, as would the exercise and conversion prices of those derivative securities.
36
The information in the following table summarizes the possible effect of the Reverse Stock
Split based upon the company’s issued and outstanding equity, as of April 1, 2016:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Split Ratio for
Issued
and Outstanding Shares
|
|
Common Stock
Outstanding after
the Reverse Stock
Split(1)
|
|
|
Warrant and
Option Shares
Reserved after the
Reverse Stock
Split(1)
|
|
|
Common Stock
Authorized after the
Reverse Stock Split
|
|
|
Post-Split
Common Stock
Authorized but
Unissued and
Unreserved after the
Reverse Stock Split(2)
|
|
|
1 for 10
|
|
|
13,922,256
|
|
|
|
9,298,862
|
|
|
|
225,000,000
|
|
|
|
201,778,882
|
|
|
1 for 11
|
|
|
12,656,596
|
|
|
|
8,453,511
|
|
|
|
225,000,000
|
|
|
|
203,889,893
|
|
|
1 for 12
|
|
|
11,601,880
|
|
|
|
7,749,051
|
|
|
|
225,000,000
|
|
|
|
205,649,069
|
|
|
1 for 13
|
|
|
10,709,427
|
|
|
|
7,152,971
|
|
|
|
225,000,000
|
|
|
|
207,137,602
|
|
|
1 for 14
|
|
|
9,944,468
|
|
|
|
6,642,044
|
|
|
|
225,000,000
|
|
|
|
208,413,488
|
|
|
1 for 15
|
|
|
9,281,504
|
|
|
|
6,199,241
|
|
|
|
225,000,000
|
|
|
|
209,519,255
|
|
|
(1)
|
As of April 1, 2016, the company had the following issued and outstanding equity: (i) 139,222,551 shares of common stock; (ii) equity awards (options and RSUs) to acquire up to 10,382,850 shares of
common stock, and (iii) warrants to acquire up to 82,605,761 shares of common stock, although approximately 23 million of these warrant shares are tied to the company’s outstanding Series B warrants, which are only exercisable if and
when the company has increased its available share capital by at least 23 million shares of common stock.
|
|
(2)
|
The actual number of shares of common stock available after the Reverse Stock Split may be higher or lower depending on the number of fractional shares that are rounded up in the Reverse Stock Split and as the result of
rounding calculations for outstanding equity awards.
|
As demonstrated in the above table, the Reverse Stock Split would, if
approved: (i) decrease the number of outstanding shares of common stock by more than 125 million shares, (ii) decrease the number of shares needed to satisfy outstanding options, RSUs and warrants by more than 85 million shares,
and (iii) maintain the company’s authorized number of shares of common stock at 225 million. As of April 1, 2016, the company had approximately 16 million authorized, but unissued and available, shares of common
stock. Consequently, the Reverse Stock Split would have the important effect of increasing the number of authorized and available shares of common stock by approximately 200 million shares, or about thirteen-fold.
In addition, all other things being equal, a reverse stock split by a publicly traded company reduces the number of shares outstanding but
leaves the market capitalization of the company the same, which should increase the price per share of the company’s stock. Put another way, after a reverse stock split, the enterprise value of the company is spread over fewer shares and so the
per share price of the stock should be commensurately higher. As an example, a hypothetical company with a market value of $50 million and 100 million shares outstanding would have a trading price of $0.50 per share ($50 million divided by 100
million), while the same company with only 25 million shares outstanding would have a trading price of $2.00 per share ($50 million divided by 25 million). We can therefore anticipate that the Reverse Stock Split would proportionately increase
the per share trading price of our outstanding common stock by an amount approximately equal to the inverse of the ratio selected by the Board (for example, an increase of 12 times current trading price for a one-for-twelve Reverse Stock Split).
Rationale for a Reverse Stock Split
The rationales for seeking approval for the Reverse Stock Split are twofold. First, as explained above, the Reverse Stock Split would increase
our available number of shares of common stock for future issuance by approximately 200 million shares. These shares could be used to raise additional capital needed to fund our operations as well as to acquire assets or other rights needed to
help grow our business, although at this time we do not have any specific plans to issue additional shares of common stock other than as disclosed in these proxy materials or otherwise in our filings with the SEC. Second, because we can anticipate
the Reverse Stock Split
37
resulting in an increase in our per share stock price, we believe it is the most appropriate way to address our previously disclosed failure to satisfy NASDAQ’s continue listing
requirements, as explained below.
Our common stock trades on the NASDAQ Capital Market, which we believe helps support and maintain stock
liquidity and company recognition for our stockholders. Companies listed on the NASAQ Capital Market, however, are subject to various rules and requirements imposed by the NASDAQ Stock Market which must be satisfied in order to continue having their
stock listed on the exchange (these are called the NASDAQ’s continued listing standards). One of these standards is the “minimum bid price” requirement, which requires that the bid price of the stock of listed companies be at least
$1.00 per share. A listed company risks being delisted and removed from the NASDAQ Capital Market if the closing bid price of its stock remains below $1.00 per share for an extended period of time.
The closing bid price of our common stock has been below $1.00 per share since April 13, 2015. On May 14, 2015, we received a
delisting determination letter from the NASDAQ Stock Market indicating that we were at risk of delisting for failure to remain in compliance with the minimum bid price standard set forth in NASDAQ Marketplace Rule 5550(a)(2). In accordance
with NASDAQ Listing Rule 5810(c)(3)(A), we had 180 calendar days, or until November 10, 2015, to regain compliance with the minimum bid price requirement. To regain compliance with the minimum bid price requirement of the NASDAQ Capital Market,
the closing bid price per share of our common stock would have to be $1.00 or higher for a minimum of ten consecutive business days during this initial 180-day compliance period. However, this did not happen, and on November 11, 2015, we
were notified by NASDAQ that we had not regained compliance with the minimum bid price requirement. NASDAQ determined that we were nevertheless eligible under NASDAQ Listing Rule 5810(c)(3)(A) for an additional 180 calendar day period, or
until May 9, 2016, to regain compliance. This second 180-day period relates exclusively to the bid price deficiency. Our common stock may be delisted during the 180 days for failure to maintain compliance with any other NASDAQ continued listing
standards during this period, such as the minimum stockholders’ equity standard among others. Our price per share and stockholders’ equity at December 31, 2015 was $0.42 and $(334,000), respectively.
If compliance cannot be demonstrated by May 9, 2016, NASDAQ will provide written notification that our common stock will be delisted. At
that time, however, we may appeal NASDAQ’s determination to a Hearings Panel. We will be asked to provide a plan to regain compliance to the Hearings Panel. Historically the Hearings Panel has generally viewed a near-term reverse stock split as
the only definitive plan acceptable to resolve a minimum bid price deficiency.
Potential Advantages of a Reverse Stock Split
Approval of this Proposal Number 3 would permit the company’s Board of Directors, in its discretion to file the First Amendment with
the Delaware Secretary of State in order to effect a reverse stock split of the company’s issued and outstanding common stock at any whole number ratio between, and inclusive of, one for ten and one for fifteen. This, we believe, could provide
a number of potential advantages, which we describe below.
Potential Advantage #1 — Effective increase in the number of
shares authorized for issuance.
Because the Reverse Stock Split would decrease the number of shares of common stock outstanding and the number of shares reserved for outstanding derivative securities, such as warrants and
options, without changing the company’s authorized capital in any way, there would be a greater proportion of shares available for issuance following the Reverse Stock Split (see the above table). We believe this effective increase in the
number of shares authorized but unissued is important to the future growth of the company because we will need to raise significant additional funds in order to advance the company’s various development programs, including continued clinical
testing of its HuCNS-SC
®
product candidate (purified human neural stem cells). These additional shares available for issuance would be used to raise money to fund the company’s working
capital and other corporate needs, for future acquisitions of assets, programs or businesses, and for other corporate purposes.
38
Adoption of the First Amendment, which would effectively increase the number of authorized
shares, would not have any immediate effect on the proportionate voting power or other rights of the existing stockholders. However, upon issuance, any additional shares of authorized common stock issued after the amendment to the restated
certificate of incorporation is effected would have rights identical to the currently outstanding shares of common stock. To the extent that the additional authorized shares of capital stock are issued in the future, they may decrease the voting
rights of existing stockholders and, depending on the price at which they are issued, could be economically dilutive to existing stockholders and have a negative effect on the market price of the common stock. Current stockholders have no preemptive
or similar rights, which means that current stockholders do not have a prior right to purchase any new issue of capital stock in order to maintain their proportionate ownership of the company. The company could also use the additional shares of
capital stock for potential strategic transactions including, among other things, acquisitions, spin-offs, strategic partnerships, joint ventures, restructurings, divestitures, business combinations, and investments, although the company has no
present plans to do so. The company cannot provide assurances that any such transactions will be consummated on favorable terms or at all, that they will enhance stockholder value or that they will not adversely affect the company’s business or
the trading price of our stock. However, we believe the effective increase in the company’s authorized capital will be important to preserving the company’s ability to opportunistically acquire assets and technologies to grow our business;
a vote against this proposal could therefore hurt our ability to grow our business and complete our existing product development efforts.
Indeed, because we believe the shares of common stock currently available for issuance by the company (approximately 16 million shares)
would be insufficient to continue operating our business as currently conducted or complete our ongoing Phase II Pathway Study in spinal cord injury, we believe the Reverse Stock Split is important for our continuing operations.
Potential Advantage #2 — Maintain NASDAQ Capital Market Listing.
We believe that having our common
stock delisted from the NASDAQ Capital Market would be undesirable for our stockholders and potentially bad for our business. Among other things, being delisted could reduce the liquidity of our common stock. We also deem valuable our ticker symbol,
which is easily recognized as “STEM” and which we could lose if we were delisted by the NASDAQ Capital Market. Also, being listed on the NASDAQ Capital Market carries with it certain prestige, and we feel it improves the recognition of our
company.
While no assurances can be given, our Board believes that a reverse stock split, at a whole number exchange ratio ranging from
one for ten (every ten shares outstanding would be combined into one share) to one for fifteen (every fifteen shares outstanding would be combined into one share), should result in an increase in the company’s price per share, and thereby help
the company meet the $1.00 per share minimum bid price requirement.
While the company’s stock price could trade above $1.00 on its
own accord over the next few months, our Board believes that it is in the company’s best interests and in the interests of our stockholders to seek approval of the proposed First Amendment to effect the Reverse Stock Split, so that we can
regain compliance even if the company’s stock trading price does not increase above $1.00 per share by May 9, 2016, the end of our second 180-day compliance period. Even if our common stock’s closing bid price were to satisfy the
minimum closing bid price requirements prior to approval of this Proposal Number 3, we may still effect the First Amendment if our stockholders approve this Proposal and our Board of Directors determines that effecting the Reverse Stock Split would
be in the best interests of the company and its stockholders.
Potential Advantage #3 — Facilitate Potential Future
Financings.
By preserving our NASDAQ Capital Market listing, we can continue to consider and pursue a wide range of future financing options to support our ongoing clinical development programs. To move our products
through the clinical, regulatory and reimbursement processes, we will need to raise additional money. We believe being listed on a national securities exchange, such as the NASDAQ Capital Market, is valued highly by many investors such as large
institutions. A listing on a national securities exchange also has the potential to create better liquidity and reduce volatility for buying and selling shares of our stock, which benefits our current and future stockholders.
39
Potential Advantage #4 — Increase Our Common Stock Price to a Level More
Appealing for Investors.
We believe that the Reverse Stock Split could enhance the appeal of our common stock to the financial community, including institutional investors, and the general investing public. We believe that
a number of institutional investors and investment funds are reluctant to invest in lower priced securities and that brokerage firms may be reluctant to recommend lower priced stock to their clients, which may be due in part to a perception that
lower-priced securities are less promising as investments, are less liquid in the event that an investor wishes to sell his, her or its shares, or are less likely to be followed by institutional securities research firms. We believe that the
reduction in the number of issued and outstanding shares of our common stock caused by the Reverse Stock Split, together with the anticipated increased stock price immediately following and resulting from the Reverse Stock Split, may encourage
further interest and trading in our common stock and thus possibly promote greater liquidity for our stockholders, thereby resulting in a broader market for our common stock than that which currently exists.
Certain Risks Associated with the Reverse Stock Split
While we believe the proposed Reverse Stock Split is critically important to the company and its stockholders, the First Amendment does carry
with it several significant risks.
We cannot assure you, for example, that the market price per share of our common stock after the
Reverse Stock Split will rise or remain constant in proportion to the reduction in the number of shares of common stock outstanding before the Reverse Stock Split. For example, using the closing price of our common stock on April 1, 2016 of
$0.26 per share as an example, if our Board of Directors were to implement the Reverse Stock Split at a one for twelve ratio, we cannot assure you that the post-split market price of our common stock would be or would remain at a price of twelve
times greater than $0.26, or $3.12 ($0.26 x 12). In some cases, the market price of a company’s shares declines after a reverse stock split. Thus, while our stock price might meet the continued listing requirements for the NASDAQ Capital Market
initially, we cannot assure you that it would continue to do so. The company completed a one for ten reverse stock split in July 2011, for example, and saw its per share price trade back below $1.00 within seven months.
The market price of our common stock will also be based on our performance and other factors, most of which are unrelated to the number of
shares outstanding. If the Reverse Stock Split is effected and the market price of our common stock declines, the percentage decline as an absolute number and as a percentage of our overall market capitalization may be greater than would occur in
the absence of a Reverse Stock Split. Furthermore, the liquidity of our common stock could be adversely affected by the reduced number of shares that would be outstanding after the Reverse Stock Split.
We also cannot assure you that the Reverse Stock Split will result in per share stock prices that will attract additional investors or
increase analyst coverage. In addition, a successful Reverse Stock Split does not guarantee the company will continue to satisfy the other continued listing requirement of the NASDAQ Capital Market.
Management is unaware of any specific effort to obtain control of the company, and has no present intention of using the proposed effective
increase in the number of authorized shares of common stock as an anti-takeover device. However, the company’s authorized, but unissued, capital stock could be used to make an attempt to effect a change in control more difficult.
Certain Risks Associated with Not Adopting the Reverse Stock Split Charter Amendment
Failure to carry out the Reverse Stock Split also carries several significant risks.
Lack of Authorized Capital; Potential Insolvency.
As of April 1, 2016, the company had 15,785,585 shares of
unissued and unreserved shares of authorized common stock. Because we currently use equity financings to
40
fund substantially all our operations, we believe that this number of authorized shares remaining would be insufficient to continue operating our business as currently conducted or complete our
ongoing Phase II Pathway Study in spinal cord injury. Failure to conduct the Reverse Stock Split therefore carries with it a high likelihood that the company will become insolvent.
Delisting.
If our stockholders do not approve the Reserve Stock Split, the company could be delisted from the
NASDAQ Capital Market, thereby potentially decreasing the liquidity of our stock and hurting or stock’s market price and discouraging future investments in our company.
Effects on Past and Future Financings.
Failure to approve this Proposal Number 3 could also have adverse
consequences on both past and future equity financings by the company. For example, the company completed in mid-March 2016 an equity financing of approximately $8 million gross proceeds in which the company issued Series B warrants
to purchase up to 22,996,747 shares of company common stock at a price per share of $0.42. These Series B warrants cannot be exercised unless the company’s stockholders approve an increase of the authorized share capital of the
company. Because the Reverse Stock Split would not change the company’s authorized capital, by approving this Proposal Number 3, our Board would be able to increase the number of common shares available for issuance by completing the
Reverse Stock Split. Consequently, the company could receive additional proceeds through the exercise of the outstanding Series B warrants, on or before their expiry in March 2022, of approximately $9.7 million. In addition, as part of
this March 2016 financing, the company agreed to solicit stockholder approval to increase the company’s authorized shares of common stock at least every four months until the company obtains stockholder approval, so failure to obtain approval
for this Proposal Number 3 at the Annual Meeting would require the company to conduct a special stockholder meeting in 2016 and incur those unanticipated costs. In addition, Proposal Number 5 (described below) describes a potential equity line
financing by the company of up to $16 million. By approving this Proposal Number 3, the company will have sufficient shares to complete the equity line financing if the company’s Board of Directors authorizes it.
Procedure for Effecting Reverse Stock Split and Exchange of Stock Certificates
Text of the First Amendment to our certificate of incorporation
. If the stockholders approve this Proposal
Number 3 to authorize our Board of Directors to implement the First Amendment and our Board of Directors decides to implement the First Amendment, we will amend Section THREE of our restated certificate of incorporation to add substantially the
following paragraph at the end thereof:
“Effective at 5:00 p.m. Pacific Time the date of filing of the Certificate of Amendment
with the State of Delaware (the “Reverse Split Effective Time”), every [ten]... [fifteen] shares of Common Stock issued and outstanding prior to the Reverse Split Effective Time, without further action, will be combined into and
automatically become one share of issued and outstanding Common Stock of the Corporation. The Corporation will not issue fractional shares on account of the foregoing Reverse Stock Split; all shares that are held by a stockholder as of the Reverse
Split Effective Time shall be aggregated and each fractional share resulting from the Reverse Stock Split after giving effect to such aggregation shall be rounded up to the nearest whole number.”
To carry out the First Amendment, we would file with the Secretary of State for the State of Delaware a certificate of amendment in
substantially the form attached as Schedule 1. Stockholders are encouraged to review this carefully as it would modify the capitalization of the company upon filing.
If this Proposal Number 3 is approved by our stockholders and if our Board of Directors concludes that the First Amendment is in the best
interests of the company and its stockholders on a date within three months after stockholder approval is obtained, our Board of Directors would cause the Reverse Stock Split to be implemented at the whole number ratio between one for ten and one
for fifteen, as selected by our Board of Directors in its sole discretion. We would file the certificate of amendment with the Delaware Secretary of State at such time as
41
our Board of Directors has determined the appropriate effective time for the First Amendment, including the Reverse Stock Split. Our Board of Directors may delay effecting the First Amendment
without resoliciting stockholder approval to any time within three months after the date stockholder approval is obtained (if at all). The First Amendment would become effective on the date the certificate of amendment is filed with the Delaware
Secretary of State (the “Reverse Split Effective Date”). Beginning on the Reverse Split Effective Date, each certificate representing Old Shares would be deemed for all corporate purposes to evidence ownership of New Shares.
As soon as practicable after the Reverse Split Effective Date, stockholders would be notified that the Reverse Stock Split has been effected.
We will retain an exchange agent (the “Exchange Agent”) for purposes of implementing the exchange of stock certificates. Holders of Old Shares would be asked to surrender to the Exchange Agent certificates representing Old Shares in
exchange for certificates representing New Shares in accordance with the procedures to be set forth in a letter of transmittal to be sent by us. No new certificates would be issued to a stockholder until such stockholder has surrendered such
stockholder’s outstanding certificate(s) together with the properly completed and executed letter of transmittal to the Exchange Agent. Any Old Shares submitted for transfer, whether pursuant to a sale or other disposition, or otherwise, would
automatically be exchanged for New Shares.
STOCKHOLDERS SHOULD NOT DESTROY ANY STOCK CERTIFICATES AND SHOULD NOT SUBMIT ANY CERTIFICATES UNTIL REQUESTED TO DO SO.
Fractional Shares
No fractional shares
would be issued in connection with the Reverse Stock Split. Stockholders of record who otherwise would be entitled to receive fractional shares, would be entitled to rounding up of their fractional share to the nearest whole share.
Effect on Options, Warrants and Other Securities
All outstanding options, warrants and other securities entitling their holders to purchase shares of our common stock would be adjusted as a
result of the Reverse Stock Split, as required by the terms of each security. In particular, the conversion ratio for each security would be reduced proportionately, and the exercise price, if applicable, would be increased proportionately, in
accordance with the terms of each security and based on the exchange ratio implemented in the Reverse Stock Split.
Accounting Matters
The First Amendment would not affect the common stock capital account on our balance sheet. As of the Reverse Split Effective Date, the stated
capital on our balance sheet attributable to our common stock would be reduced proportionately based on the selected exchange ratio, and the additional paid-in capital account would be credited with the amount by which the stated capital is reduced.
In future financial statements, we would restate net income or loss per share and other per share amounts for periods ending before the Reverse Stock Split to give retroactive effect to the Reverse Stock Split. The per share net income or loss and
net book value of our common stock would be increased because there would be fewer shares of our common stock outstanding.
Discretionary Authority of
the Board of Directors to Abandon Reverse Stock Split
The Board of Directors reserves the right to abandon the First Amendment without
further action by our stockholders at any time before the effectiveness of the filing with the Delaware Secretary of State of the certificate of amendment to the company’s certificate of incorporation, even if the Reverse Stock Split has been
authorized by our stockholders at the Annual Meeting. By voting in favor of the Reverse Stock Split, you are expressly also authorizing our Board of Directors to determine not to proceed with, and abandon, the Reverse Stock Split, if it should so
decide.
42
No Dissenter’s Rights
Neither Delaware law, the company’s restated certificate of incorporation, nor the company’s amended and restated by-laws provides
for appraisal or other similar rights for dissenting stockholders in connection with this proposal. Accordingly, the company’s stockholders will have no right to dissent and obtain payment for their shares, and we will not independently provide
stockholders with any such right.
Directors’ Recommendation
The affirmative vote of a majority of all outstanding shares of common stock is required to approve the Reverse Stock Split at the Annual
Meeting. If you abstain from voting on this Proposal Number 3 to approve the Reverse Stock Split, it will have the same effect as a vote “AGAINST” the proposal. Your vote is therefore extremely important.
THE BOARD OF DIRECTORS RECOMMENDS A VOTE
FOR
THE PROPOSAL TO AMEND THE COMPANY’S CERTIFICATE OF INCORPORATION TO EFFECT THE REVERSE STOCK SPLIT
OF THE COMPANY’S ISSUED AND OUTSTANDING COMMON STOCK AS DESCRIBED IN THIS PROPOSAL NUMBER 3.
Please Note:
This proxy statement contains
forward looking statements within the meaning of Section 27A of the Securities Act and Section 21E of the Securities Exchange Act that involve substantial risks and uncertainties. Such statements include, without limitation, all statements
as to expectation or belief and statements as to the future trading price and liquidity of our common stock, whether traded on the NASDAQ Capital Market or otherwise; our future results of operations; our ability to satisfy all the listing
requirements of NASDAQ; the prospect for analyst coverage; and the need for, and timing of, additional capital and capital expenditures. Our actual results may vary materially from those contained in such forward-looking statements because of risks
to which we are subject, including the fact that uncertainties in our ability to obtain the capital resources needed to continue our current research and development operations and to conduct the research, preclinical development and clinical trials
necessary for regulatory approvals; the uncertainty regarding our ability to obtain a corporate partner or partners, if needed, to support the development and commercialization of our potential cell-based therapeutics products; the uncertainty
regarding the outcome of our clinical trials or studies we may conduct in the future; the uncertainty regarding the validity and enforceability of our issued patents; the uncertainty regarding whether any products that may be generated in our
cell-based therapeutics programs will prove clinically safe and effective; the uncertainty regarding whether we will achieve significant revenue from product sales or become profitable; competition from third parties; intellectual property rights of
third parties; litigation risks; and other risks to which we are subject. All forward-looking statements attributable to us or to persons acting on our behalf are expressly qualified in their entirety by the cautionary statements and risk factors
set forth in “Risk Factors” in Part I, Item 1A of our Form 10-K for the fiscal year ended December 31, 2015.
43
PROPOSAL NUMBER 4
Approval of Amendment to the Company’s
Certificate of Incorporation to Decrease Authorized Capital
General
Our Board of Directors has
unanimously adopted a resolution declaring advisable and recommending to the stockholders for their approval a proposal to amend the company’s restated certificate of incorporation, as amended to date, to decrease the number of authorized
shares of common stock from 225,000,000 to 200,000,000. Approval of this Proposal Number 4 would grant our Board the authority, without further action by the stockholders, to decrease the authored capital of the company, by decreasing the number of
authorized shares of common stock from 225 million to 200 million, at any time within three months after the date stockholder approval for this proposal to decrease the company’s authorized capital is obtained. As explained below, our
Board’s decision whether or not (and when) to decrease the authorized capital of the company would be based on a number of factors, including whether the Company effects the Reverse Stock Split described in Proposal Number 3.
A sample form of the certificate of amendment relating to this Proposal Number 4, which we would file with the Secretary of State of the State
of Delaware to carry out the amendment to our certificate of incorporation, is attached to this proxy statement as Schedule 2 (the “Second Amendment”).
Rationale for a Reduction in the Number of Shares of Authorized Common Stock
We do not intend to file the Second Amendment unless our stockholders approve Proposal Number 3 and we effect the Reverse Stock Split, as
described in Proposal Number 3. Indeed, we believe Delaware law precludes us from filing the Second Amendment, which would reduce the number of authorized shares of common stock to 200 million, unless we first complete the Reverse Stock Split,
because the company already has more than 200 million shares of common stock issued and outstanding on a fully diluted basis. We therefore expect that a reduction in authorized capital at this time would be contingent upon the successful
completion of a Reverse Stock Split, as described above in Proposal Number 3. If the stockholders approve Proposal Number 3 and if the Board chooses to complete a Reverse Stock Split, we may elect to file the Second Amendment to decrease our
authorized common stock to 200 million, if our Board believes that we have sufficient authorized and available shares of common stock, given our trading price after the Reverse Stock Split, to fund our foreseeable operations.
Our Board will only file the Second Amendment to decrease the company’s authorized capital if it believes the 200 million shares of
authorized common stock, after the Reverse Stock Split and given the per share price of the outstanding common stock, is sufficient for the company’s financing and product development needs.
44
Effects of a Reverse Stock Split
We described, under Proposal Number 3, the anticipated impact on our authorized share capital as a result of the proposed Reverse Stock Split.
Because the Reverse Stock Split would not change the company’s authorized shares, a Reverse Stock Split would increase the number of shares of common stock available to us for future use, as shown in the following table, which summarizes the
possible effect of the Reverse Stock Split based upon the company’s issued and outstanding equity, as of April 1, 2016:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Split Ratio for
Issued
and Outstanding Shares
|
|
Common Stock
Outstanding after
the Reverse Stock
Split(1)
|
|
|
Warrant and
Option Shares
Reserved after the
Reverse Stock
Split(1)
|
|
|
Available
Common Stock
after the Reverse
Stock Split, if
225 million
shares
authorized(2)
|
|
|
Available
Common Stock
after the Reverse
Stock Split, if
200 million shares
authorized(2)
|
|
|
1 for 10
|
|
|
13,922,256
|
|
|
|
9,298,862
|
|
|
|
201,778,882
|
|
|
|
176,778,882
|
|
|
1 for 11
|
|
|
12,656,596
|
|
|
|
8,453,511
|
|
|
|
203,889,893
|
|
|
|
178,889,893
|
|
|
1 for 12
|
|
|
11,601,880
|
|
|
|
7,749,051
|
|
|
|
205,649,069
|
|
|
|
180,649,069
|
|
|
1 for 13
|
|
|
10,709,427
|
|
|
|
7,152,971
|
|
|
|
207,137,602
|
|
|
|
182,137,602
|
|
|
1 for 14
|
|
|
9,944,468
|
|
|
|
6,642,044
|
|
|
|
208,413,488
|
|
|
|
183,413,488
|
|
|
1 for 15
|
|
|
9,281,504
|
|
|
|
6,199,241
|
|
|
|
209,519,255
|
|
|
|
184,519,255
|
|
|
(1)
|
As of April 1, 2016, the company had the following issued and outstanding equity: (i) 139,222,551 shares of common stock; (ii) equity awards (options and RSUs) to acquire up to 10,382,850 shares of
common stock, and (iii) warrants to acquire up to 82,605,761 shares of common stock, although approximately 23 million of these warrant shares are tied to the company’s outstanding Series B warrants, which were issued in the
company’s March 2016 financing and are only issuable if and when the company is able to increase its authorized share capital by at least 23 million shares of common stock.
|
|
(2)
|
The actual number of shares of common stock available after the Reverse Stock Split may be higher or lower depending on the number of fractional shares that are rounded up in the Reverse Stock Split and as the result of
rounding calculations for outstanding equity awards.
|
If our per share trading price remains stable following the Reverse
Stock Split and adjusts proportionally, then we could expect to have a sufficient number of shares of common stock to raise over $600 million (approximately 200 million available shares multiplied by a per share price of approximately $3.00).
However, the market price of our common stock will be based on many factors, including our clinical trial results, business performance and market factors, almost all of which are completely unrelated to the number of shares outstanding. If the
Reverse Stock Split is effected and the market price of our common stock declines, the percentage decline as an absolute number and as a percentage of our overall market capitalization may be greater than would occur in the absence of a Reverse
Stock Split. Furthermore, the liquidity of our common stock could be adversely affected by the reduced number of shares that would be outstanding after the Reverse Stock Split.
Principal Effects of the Second Amendment
Text of the Second Amendment to our certificate of incorporation.
If the stockholders approve this Proposal
Number 4 to authorize our Board of Directors to implement the Second Amendment and our Board of Directors decides to implement the Second Amendment, we will amend the first sentence of Section THREE of our restated certificate of incorporation
to read as follows:
“The total number of shares of capital stock which the Corporation shall have the authority to issue shall be
201,000,000 shares, consisting of (i) 200,000,000 shares of Common Stock and (ii) 1,000,000 shares of Undesignated Preferred Stock.”
To carry out the Second Amendment, we would file with the Secretary of State for the State of Delaware a certificate of amendment in
substantially the form attached as Schedule 2. Stockholders are encouraged to review this carefully as it would modify the capitalization of the company upon filing.
45
Effect on Options, Warrants and Other Securities
The Second Amendment would have no effect on our outstanding common stock, options, warrants, and other equity securities.
Accounting Matters
The Second Amendment
would not affect the common stock capital account on our balance sheet.
Discretionary Authority of the Board of Directors to Abandon Decrease of
Authorized Capital
The Board of Directors reserves the right to abandon the Second Amendment without further action by our
stockholders at any time before the effectiveness of the filing with the Delaware Secretary of State of the certificate of amendment to the company’s certificate of incorporation decreasing the authorized capital, even if the reduction in
authorized capital has been authorized by our stockholders at the Annual Meeting. By voting in favor of reducing the number of authorized shares of common stock to 200 million, you are expressly also authorizing our Board of Directors to
determine not to proceed with, and abandon, this reduction in the authorized capital, if it should so decide, and irrespective of our per share trading price.
No Dissenter’s Rights
Neither
Delaware law, the company’s restated certificate of incorporation, nor the company’s amended and restated by-laws provides for appraisal or other similar rights for dissenting stockholders in connection with this proposal. Accordingly, the
company’s stockholders will have no right to dissent and obtain payment for their shares, and we will not independently provide stockholders with any such right.
Directors’ Recommendation
The
affirmative vote of a majority of all outstanding shares of common stock is required to approve at the Annual Meeting the proposed decrease in the number of authorized shares of common stock to 200 million. If you abstain from voting on this
Proposal Number 4 to approve the proposal to reduce the company’s authorized capital, it will have the same effect as a vote “AGAINST” the proposal. Your vote is therefore extremely important.
THE BOARD OF DIRECTORS RECOMMENDS A VOTE
FOR
THE PROPOSAL TO AMEND THE COMPANY’S CERTIFICATE OF INCORPORATION TO DECREASE THE NUMBER OF
AUTHORIZED SHARES OF COMMON STOCK TO 200 MILLION AS DESCRIBED IN THIS PROPOSAL NUMBER 4.
46
PROPOSAL NUMBER 5
Approval of $16 Million Equity Line Financing
We are asking our stockholders to approve the sale by the company of up to $16 million in company common stock to a single institutional
investor (an “Investor”), in accordance with a committed equity line purchase agreement (a “Purchase Agreement”), as further described below, at prices that may be less than the greater of book or market value of the
company’s common stock at the date we execute the Purchase Agreement. The Board believes it is in the best interests of the company and our stockholders for the company to have the ability to sell shares of our common stock under this equity
line even if at prices per share below the trading price when we enter into the Purchase Agreement.
Nasdaq Requirements
Because our common stock is listed on the NASDAQ Capital Market, we are subject to NASDAQ Listing Rules. NASDAQ Listing Rule 5635 requires
that a listed company obtain stockholder approval in connection with a transaction, other than a public offering, if involving the potential issuance of common stock (or securities convertible into or exercisable for common stock) equal to 20% or
more of its common stock or 20% or more of its voting power outstanding before the issuance, for less than the greater of book or market value of the stock as of the date of the transaction. In addition, NASDAQ Listing Rule 5635 requires
shareholder approval prior to the sale or issuance or potential issuance of common stock, in a transaction that would result in a change in control. Published NASDAQ interpretative guidance indicates that the ownership by a person of 20% or more of
the outstanding common stock as a result of the issuance or potential issuance of shares could result in a change in control requiring shareholder approval.
As proposed, the equity line to be established by the Purchase Agreement could result in the issuance of 20% or more of our common stock to
the Investor for less than the greater of book value or market value as of the date we enter into the Purchase Agreement. Sales under the equity line could also result in a change of control for purposes of NASDAQ Listing Rule 5635, even though we
expect the Purchase Agreement would limit the Investor to holding no more than 9.99% of our outstanding shares. Therefore, to comply with NASDAQ Listing Rule 5635, we are seeking stockholder approval to issue up to $16 million in common stock to the
Investor, at an average sales price which could be lower than the market price for our stock on the date we enter into the Purchase Agreement, for the reasons set forth below.
Purpose and Background of the Equity Line
Based on current market prices, if we were to sell all of the shares of common stock available to us to the Investor in a private offering
without shareholder approval, it would result in total proceeds to us of approximately $6.8 million (i.e., 19.99% of our public float), far less than the $16 million we would like to be able to access under the Purchase Agreement. Moreover, our
stock price has fluctuated significantly over the past 12 months and could continue to experience significant fluctuations, which would affect the amount of proceeds available to us under any equity line agreement. Therefore, we are seeking
shareholder approval to issue shares of our common stock worth up to $16 million under the proposed equity line, as more fully described below, to give us the flexibility to exceed the 20% cap if we believe doing so is in the best interests of the
company and its stockholders. The extent to which we will be able to draw down, or will need to draw down, under the proposed equity line agreement would be dependent upon various factors, including our future financial performance and the market
price of our common stock at the time of any put exercise, as described below.
47
Terms of the Proposed Transaction
At present, we do have not have an existing equity line agreement with any particular investor. If we obtain stockholder approval for the
Equity Line arrangement, as described in this proposal, we expect to endeavor to secure an equity line commitment from an institutional investor with experience providing equity line arrangements to companies in our industry, such as Azimuth
Opportunity, Crede Capital Group, Lincoln Park Capital, Redland Capital Partners, and Tarpon Bay Partners. We would expect the terms of any such equity line arrangement to be substantially similar to those described herein. However, we can
give no assurance that we will be able to secure an equity line arrangement on substantially these terms, if at all. Final terms of the Equity Line, assuming one is entered into by the company within three months following our annual
stockholder meeting, may be different than those described here. If our Board elects to enter into a different type of financing transaction or an equity line financing on substantially different terms than those described here, stockholder
approval of this Proposal Number 5 may not satisfy any stockholder vote standards under the Nasdaq Capital Market’s continued listing requirements.
Equity Line Purchase Shares
The
company is contemplating entering into the equity line Purchase Agreement with the Investor, as a source of liquidity for the company, under which we would have the right to sell, and the Investor would be obligated to purchase, shares of our common
stock worth up to $16 million in aggregate purchase price (the “Purchase Shares”) in the form of an equity line (“Equity Line ”) over an expected 24-month period commencing on the date that a resale registration statement
registering the Purchase Shares (the “Registration Statement”) is declared effective by the SEC and a final prospectus in connection therewith is filed with the SEC (the “Commencement Date”). We would enter into the Purchase
Agreement, if at all, within three months after stockholder approval for this Proposal Number 4 is obtained.
On the Commencement
Date, the Investor would purchase an initial number of shares at a price per share expected to equal the lesser of: (a) the closing price the day prior to the Commencement Date or (b) the average closing price for an agreed upon number of
business days prior to the Commencement Date. In addition, as consideration for entering into the Purchase Agreement, as described above, we would expect to issue to Investor common stock with a dollar value, as of the Commencement Date, equal to
approximately 2% of the total dollar amount of the Equity Line (the “Commitment Shares”), at a calculated price to be based on the average of the closing sale prices of our common stock for a specified period prior to the Commencement
Date.
After this initial purchase, we would be able to direct the Investor, at our election and subject to certain conditions, to
purchase the remaining Purchase Shares, for an aggregate purchase price under the Equity Line of up to the full $16 million, pursuant to agreed-upon procedures between the company and the Investor. At any time after the Commencement Date, the
company would have the right to direct the Investor to buy a number of shares of common stock at purchase price calculated based on a pricing formula based on sale prices on or prior to the date the company provides notice to the Investor of the
transaction. As the trading price of our common stock increases, the number of shares the company would be able to sell on any given date would increase. We expect the company would be able to submit purchase notices to the Investor as often as
every other business day, subject to an agreed-upon dollar-based cap.
We would have no obligation to issue any minimum number of Purchase
Shares pursuant to the Equity Line, with the exception of the Commitment Shares described below. The company would have the right to terminate the Equity Line, at its sole election, at any time.
48
Limitation on Issuance
As proposed, the Purchase Shares could not be issued if, after giving effect to the issuance, the Investor would beneficially own in excess of
9.99% of the outstanding shares of our common stock.
Registration Rights
Upon obtaining shareholder approval, we would file the Registration Statement covering the resale of the Purchase Shares and the Commitment
Shares, up to the aggregate maximum of $16 million, as specified herein.
Other Anticipated Provisions
We can anticipate other provisions to apply in the equity line Purchase Agreement, including before we could sell shares to the Investor:
|
|
•
|
|
our common stock would need to be listed on the NASDAQ Capital Market or on an equivalent national securities exchange;
|
|
|
•
|
|
no injunction or other governmental action shall remain in force prohibiting the purchase by, or the issuance of the Purchase Shares to the Investor; and
|
|
|
•
|
|
our representations and warranties to the Investor must be true and correct in all material respects.
|
We would also expect the equity line Purchase Agreement to contain customary representations, warranties and indemnification provisions.
As part of the agreement, the Investor would agree not to sell short any of our securities, either directly or indirectly through its
affiliates, principals or advisors during the term of the Equity Line. However, in connection with the distribution of our common stock or otherwise, the Investor could enter into certain other hedging transactions. In addition, the Investor could
sell some, all or none of its shares of our common stock. We currently have no agreements, arrangements or understandings with any investor regarding the sale or other disposition of any of the Purchase Shares, nor would we expect to have any such
agreements, arrangements or understandings as part of the equity line Purchase Agreement. However, we currently expect the Investor would promptly resell all shares that we sell to the Investor as part of the Equity Line.
Use of Proceeds
We intend to use
any net proceeds raised through the Equity Line for general corporate purposes, including working capital, capital expenditures, research and development expenditures and clinical trial expenditures. A portion of the net proceeds may also be used
for the acquisition of businesses, products and technologies that are complementary to ours, or for other strategic purposes, although we have no current understandings, commitments or agreements to do so. Pending use of the net proceeds, we intend
to invest the net proceeds in short-term, interest-bearing, investment-grade securities.
Effect on Currently Outstanding Common Stock
Our stockholders have no preemptive rights entitling them to purchase a pro rata share of the common stock sold pursuant to the Equity
Line. Therefore, the issuance of shares of our common stock pursuant to the Equity
49
Line could be significantly dilutive in voting power to our existing stockholders. In addition, the market price of our common stock could decline as a result of our utilization of the Equity
Line.
If Proposal No. 5 is adopted, we would be able to issue shares of common stock pursuant to the Equity Line, as described
above, including the Purchase Shares and the Commitment Shares, at a price less than the greater of book or market value of the company’s common stock at the date of execution of the Purchase Agreement. The Purchase Shares and Commitment Shares
may also be sold by us at a price per share below today’s trading price and below the price per share on the Commencement Date. Consequently, the issuance of these shares could result in significant dilution to our stockholders. Additionally,
the sale or any resale into the public markets of the common stock issued in connection with any equity line purchase agreement could cause the market price of our common stock to decline.
In addition, we have outstanding warrants to acquire up to 82,605,761 shares of our common stock at an exercise price per share (in other
words, a “strike price” per share) ranging from $0.2999998 to $1.80, meaning that these warrant holders can elect, until the expiry of their warrants, to purchase additional shares of our common stock at a price ranging from $0.2999998 to
$1.80 per share. However, approximately half of these warrants contain “full ratchet” price protection, which means that the per share strike price for these warrants can be reset, under certain circumstances, to a lower per share strike
price. Most commonly, this occurs when the company issues additional shares of common stock in a financing at an imputed per share price below the warrants’ then-existing per share strike price. Doing so results in the warrants’ per share
strike price adjusting down to the per share price of the newly issued shares.
An adjustment to the per share strike price could result
in substantial additional dilution to our existing stockholders. For example, as of April 1, 2016, we had the following outstanding warrants with full ratchet price protection:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Description of Warrants
|
|
Total Shares
Underlying
the Warrants
|
|
|
Current Strike
Price (per shr)
|
|
|
Warrant Expiration
Date
|
|
|
Series A Warrants (December 2011 financing)
|
|
|
6,936,880
|
|
|
$
|
0.2999998
|
|
|
|
Dec 2016
|
|
|
Series A Warrants (March 2016 financing)
|
|
|
15,331,165
|
|
|
$
|
0.2999998
|
|
|
|
Mar 2018
|
|
|
Series B Warrants (March 2016 financing)
|
|
|
22,996,747
|
|
|
$
|
0.42
|
|
|
|
Mar 2022
|
|
Therefore, if we were to sell additional shares of common stock under the Equity Line at a price per
share below $0.42 per share but above $0.30 per share, warrants to acquire up to approximately 22 million additional shares of common stock would have their strike price lowered to the sales price under the Equity Line. Similarly, if we were to
sell additional shares of common stock under the Equity Line at a price per share below $0.2999998 per share, then warrants entitling holders to purchase up to approximately 45 million additional shares of common stock would reset to the lower per
share purchase price of the sale under the Equity Line.
Required Vote
The affirmative vote of a majority of the Company’s common stock voted at the Annual Meeting, by person or by proxy, is
required to approve this Proposal Number 5. Broker non-votes and abstentions will not be treated as votes cast on the proposal. Unless otherwise directed by marking the accompanying proxy, the proxy holders will vote “FOR” the
proposal to authorize the Company to issue up to $16 million of common stock pursuant to the Equity Line.
50
A vote against this Proposal Number 5 would not prevent the company from entering into
an equity line agreement with the Investor on substantially the terms described above. However, without stockholder approval, the company would be precluded from selling through the Equity Line more than an amount equal to 19.99% of its shares
outstanding, calculated as of the Commencement Date, unless the average price of all sales under the Equity Line are above the per share trading price of our stock on the Commencement Date. We believe this price floor limitation could
substantially reduce the utility of the proposed Equity Line and accordingly:
THE BOARD OF DIRECTORS RECOMMENDS THAT STOCKHOLDERS VOTE
FOR
THE PROPOSAL TO APPROVE THE SALE BY THE COMPANY OF UP TO $16 MILLION IN COMPANY COMMON STOCK, IN ACCORDANCE WITH A COMMITTED EQUITY LINE PURCHASE AGREEMENT, AS FURTHER DESCRIBED ABOVE, AT PRICES THAT MAY BE LESS THAN THE GREATER OF BOOK OR
MARKET VALUE OF THE COMPANY’S COMMON STOCK AT THE DATE OF EXECUTION OF THE AGREEMENT.
51
OTHER MATTERS
Stockholder Proposals
Stockholders who
wish to present proposals for inclusion in the company’s proxy materials for the 2017 Annual Meeting of Stockholders may do so by following the procedures prescribed in Rule 14a-8 under the Exchange Act. To be eligible, the stockholder
proposals must be received by our corporate secretary on or before November 23, 2016.
Stockholders who wish to make a proposal at the
2017 Annual Meeting of Stockholders, other than one that will be included in our proxy materials, must notify us no later than February 6, 2017 (see Rule 14a-4 under the Exchange Act). If a stockholder who wishes to present a proposal at the
2017 Annual Meeting of Stockholders fails to notify us by February 6, 2017, the proxies that management solicits for the meeting will confer discretionary authority to vote on the stockholder’s proposal if it is properly brought before the
meeting.
Stockholder Nominations of Directors
Stockholders or groups of stockholders that, individually or as a group, have beneficially owned at least 5% of the company’s common
stock for at least one year prior to the date of such submission (the “Nominating Stockholder”) may submit a candidate for nomination for election as a director at any annual meeting of stockholders in accordance with Board policy. The
submission must be in writing and delivered to StemCells, Inc., Attn: Secretary, Board of Directors, 7707 Gateway Blvd., Newark, California 94560, no later than on or about November 23, 2016 for nominees to be considered for nomination at the 2017
annual meeting. Submissions must include the name, address and number of shares of common stock beneficially owned by each participant in the Nominating Stockholder group, a representation that the Nominating Stockholder meets the requirements
described in the Board policy and will continue to meet them through the date of the annual meeting, a description of all arrangements or understandings between or among the Nominating Stockholder group (or any participant in the Nominating
Stockholder group) and the candidate or any other person or entity regarding the candidate, all information regarding the candidate that the company would be required to disclose in a proxy statement under SEC rules, including whether the candidate
is independent or, if not, a description of the reasons why not, the consent of the candidate to serve as a director, and representations by the candidate regarding his or her performance of the duties of a director. Full details may be obtained
from the secretary of the Board of Directors at the address above or on our website at www.stemcellsinc.com. The Corporate Governance Committee will consider and evaluate up to two candidates recommended in accordance with this policy in connection
with any annual meeting. The Corporate Governance Committee will consider and evaluate candidates recommended by stockholders on the same basis as candidates recommended by other sources.
In addition, the company’s by-laws provide that a stockholder entitled to vote for the election of directors at a meeting may nominate
persons for election as directors by giving timely notice thereof in proper written form to the Secretary accompanied by a petition signed by at least 100 record holders of capital stock of the company representing in the aggregate 1% or more of the
outstanding shares entitled to vote in the election of directors, which petition must show the class and number of shares held by each person. To be timely, such notice and petition must be received at the principal executive offices of the company
not less than 60 days nor more than 90 days prior to the meeting, except if less than 70 days notice of the date of the meeting is given to stockholders, in which case the notice and petition must be received not later than the close
of business on the tenth day following the day on which notice of the date of the meeting was mailed or public disclosure of such date was made. The requesting stockholder is required to provide information with respect to the nominee(s) for
director similar to that described above, as more fully set forth in the company’s by-laws.
52
Form 10-K
The company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2015, as amended, as filed with the SEC, is
available without charge upon request by writing to StemCells, Inc. at 7707 Gateway Blvd., Newark, California 94560, Attention: Investor Relations. A copy of this report is also available through our website at www.stemcellsinc.com or,
alternatively, at www.sec.gov.
“Householding” of Proxy Materials
The SEC has adopted rules that permit companies and intermediaries such as brokers to satisfy delivery requirements for proxy statements with
respect to two or more stockholders sharing the same address by delivering a single proxy statement addressed to those stockholders. This process, which is commonly referred to as “householding,” potentially provides extra convenience for
stockholders and cost savings for companies. The company and some brokers household proxy materials, delivering a single proxy statement to multiple stockholders sharing an address unless contrary instructions have been received from the affected
stockholders. Once you have received notice from your broker or us that they or we will be householding materials to your address, householding will continue until you are notified otherwise or until you revoke your consent. If, at any time, you no
longer wish to participate in householding and would prefer to receive a separate proxy statement, or if you are receiving multiple copies of the proxy statement and wish to receive only one, please notify your broker if your shares are held in a
brokerage account or us if you hold registered shares. We undertake to deliver promptly upon any request a separate or single copy of the proxy materials. You can notify us by sending a written request to StemCells, Inc., 7707 Gateway Blvd., Newark,
California 94560, Attention: Investor Relations or by calling us at (510) 456-4000.
Other Business
The Board of Directors knows of no business that will come before the meeting for action except as described in the accompanying Notice of
Meeting. However, as to any such business, the persons designated as proxies will have authority to act in their discretion.
|
|
|
By Order of the Board of Directors
|
|
|

|
|
|
|
Kenneth B. Stratton, J.D.
|
|
Secretary
|
April 8, 2016
53
Schedule I
CERTIFICATE OF AMENDMENT TO THE
RESTATED CERTIFICATE OF INCORPORATION OF
STEMCELLS, INC.
StemCells, Inc. (the “Corporation”), a corporation duly organized and existing under the General Corporation Law of the State of
Delaware (the “DGCL”), hereby certifies that:
|
1.
|
Upon the filing and effectiveness (the “Effective Time”) pursuant to the DGCL and this Certificate of Amendment of the Restated Certificate of Incorporation of the Corporation, as amended (the
“Charter”), each [ten to fifteen] shares of the Corporation’s common stock, par value $.001 per share, issued and outstanding immediately prior to the Effective Time (the “Old Shares”) shall automatically be combined into
one validly issued, fully paid and non-assessable share of common stock without any further action by the Corporation or the holder thereof, subject to the treatment of fractional share interests as described below (the “Reverse Stock
Split”). The Corporation shall not issue fractional shares in connection with the Reverse Stock Split. Holders of Old Shares who would otherwise be entitled to receive a fraction of a share on account of the Reverse Stock Split shall have their
fractional share rounded up to the nearest whole number as of the Effective Time.
|
|
2.
|
This Certificate of Amendment shall become effective on [
], 2016 at 12:01 a.m. Eastern Time.
|
|
3.
|
This Certificate of Amendment was duly adopted in accordance with Section 242 of the DGCL. The Board of Directors duly adopted resolutions setting forth and declaring advisable this Certificate of Amendment and
directed that the proposed Amendment be considered by the stockholders of the Corporation. A meeting of stockholders was duly called upon notice in accordance with Section 222 of the DGCL and held on May [5], 2016, at which meeting the
necessary number of shares were voted in favor of the proposed amendment. The stockholders of the Corporation duly adopted this Certificate of Amendment.
|
|
4.
|
The other provisions of the Charter are not affected by the aforementioned amendment and remain in full force and are not affected by this Certificate of Amendment.
|
IN WITNESS WHEREOF, StemCells, Inc. has caused this Certificate of Amendment to be signed by its President and Chief Executive Officer, Ian
Massey, and its corporate Secretary, Kenneth B. Stratton, on this [
day of
], 2016.
|
|
|
STEMCELLS, INC.
|
|
|
|
|
|
Ian Massey
|
|
President and Chief Executive Office
|
|
|
|
|
|
Kenneth B. Stratton
Secretary
|
I-1
Schedule II
CERTIFICATE OF AMENDMENT TO THE
RESTATED CERTIFICATE OF INCORPORATION OF
STEMCELLS, INC.
StemCells, Inc. (the “Corporation”), a corporation duly organized and existing under the General Corporation Law of the State of
Delaware (the “DGCL”), hereby certifies that:
|
1.
|
Upon the Effective Time, the first paragraph of section THREE of the Charter is hereby amended to read in its entirety as set forth below:
|
The total number of shares of stock that this Corporation shall have authority to issue is 201,000,000, consisting of 200,000,000 shares of
Common Stock, with a par value of $0.01 per share (the “Common Stock”), and 1,000,000 shares of Undesignated Preferred Stock with a par value of $0.01 per share (the “Undesignated Preferred Stock”).
|
2.
|
This Certificate of Amendment shall become effective on [
], 2016 at 12:01 a.m. Eastern Time.
|
|
3.
|
This Certificate of Amendment was duly adopted in accordance with Section 242 of the DGCL. The Board of Directors duly adopted resolutions setting forth and declaring advisable this Certificate of Amendment and
directed that the proposed Amendment be considered by the stockholders of the Corporation. A meeting of stockholders was duly called upon notice in accordance with Section 222 of the DGCL and held on May [5], 2016, at which meeting the
necessary number of shares were voted in favor of the proposed amendment. The stockholders of the Corporation duly adopted this Certificate of Amendment.
|
|
4.
|
The remaining provisions of the Charter, including without limitation the remaining provisions of section THREE, are not affected by the aforementioned amendment and remain in full force and are not affected by this
Certificate of Amendment.
|
IN WITNESS WHEREOF, StemCells, Inc. has caused this Certificate of Amendment to be signed by
its President and Chief Executive Officer, Ian Massey, and its corporate Secretary, Kenneth B. Stratton, on this [
day of
], 2016.
|
|
|
STEMCELLS, INC.
|
|
|
|
|
|
Ian Massey
|
|
President and Chief Executive Office
|
|
|
|
|
|
Kenneth B. Stratton
Secretary
|
II-1

STEMCELLS
STEMCELLS, INC.
C/O EQUISERVE TRUST COMPANY N.A.
P.O. BOX 8694
EDISON, NJ 08818-8694
VOTE BY INTERNET - www.proxyvote.com
Use the Internet to transmit your voting instructions and for electronic delivery of information up until 11:59 P.M. Eastern Time the day before the meeting date. Have your proxy
card in hand when you access the web site and follow the instructions to obtain your records and to create an electronic voting instruction form.
ELECTRONIC
DELIVERY OF FUTURE PROXY MATERIALS
If you would like to reduce the costs incurred by our company in mailing proxy materials, you can consent to receiving all
future proxy statements, proxy cards and annual reports electronically via e-mail or the Internet. To sign up for electronic delivery, please follow the instructions above to vote using the Internet and, when prompted, indicate that you agree to
receive or access proxy materials electronically in future years.
VOTE BY PHONE - 1-800-690-6903
Use any touch-tone telephone to transmit your voting instructions up until 11:59 P.M. Eastern Time the day before the meeting date. Have your proxy card in hand when you call and
then follow the instructions.
VOTE BY MAIL
Mark, sign and date your proxy
card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717.
Investor
Address Line 1
Investor Address Line 2
Investor Address Line 3
Investor Address Line 4
Investor Address Line 5
John Sample
1234 ANYWHERE STREET
ANY CITY, ON A1A 1A1
1 OF 2 1 1
NAME
THE COMPANY NAME INC. - COMMON
THE COMPANY NAME INC. - CLASS A
THE COMPANY NAME INC. - CLASS B
THE COMPANY NAME INC. - CLASS C
THE COMPANY NAME INC. - CLASS D
THE COMPANY NAME INC. - CLASS E
THE COMPANY NAME INC. - CLASS F
THE COMPANY NAME INC. - 401 K
CONTROL # 0000000000000000
SHARES 123,456,789,012.12345
123,456,789,012.12345
123,456,789,012.12345
123,456,789,012.12345
123,456,789,012.12345
123,456,789,012.12345
123,456,789,012.12345
123,456,789,012.12345
PAGE 1 OF 2
TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: [X]
KEEP THIS PORTION FOR YOUR RECORDS DETACH AND RETURN THIS PORTION ONLY
THIS PROXY CARD IS
VALID ONLY WHEN SIGNED AND DATED.
To withhold authority to vote for any individual nominee(s), mark “For All Except” and write the number(s) of the
nominee(s) on the line below.
For All Withhold All For All Except
02
0000000000
The Board of Directors recommends you vote FOR the following:
1.
Election of Directors
Nominees
01 Eric Bjerkholt 02 R. Scott Greer
The Board of Directors recommends you vote FOR proposals 2, 3, 4 and 5.
For
Against Abstain
2 To ratify the selection of Grant Thornton LLP as independent public accountants for the company for the fiscal year ending December 31, 2016.
3 To approve an amendment to the company’s certificate of incorporation to effect a reverse stock split of the company’s issued and outstanding common
stock, as described in the accompanying proxy statement.
4 To approve an amendment to the company’s certificate of incorporation to reduce the number of
authorized shares of common stock to 200,000,000.
5 To approve the sale by the company of up to $16 million in company common stock, in accordance with a committed
equity line purchase agreement, as described in the accompanying proxy statement.
NOTE: Such other business as may properly come before the meeting or any
adjournment thereof.
Please sign exactly as your name(s) appear(s) hereon. When signing as attorney, executor, administrator, or other fiduciary, please give full
title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or partnership name, by authorized officer.
For address change/comments, mark here. (see reverse for instructions)
0000284697_1 R1.0.1.25
Signature [PLEASE SIGN WITHIN BOX]
Date JOB #
Signature (Joint Owners)
Date SHARES CUSIP # SEQUENCE #

Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting: The Notice & Proxy Statement, Form 10-K
is/are available at www.proxyvote.com
STEMCELLS, INC.
ANNUAL MEETING OF
STOCKHOLDERS, May 5, 2016
THIS PROXY IS SOLICITED ON BEHALF OF THE BOARD OF DIRECTORS
The undersigned stockholder, by completing this card, hereby appoints Ian Massey and Kenneth Stratton, or either of them with power of substitution to each, proxies of the
undersigned to vote at the Annual Meeting of Stockholders of StemCells, Inc. to be held on May 5, 2016 at 7707 Gateway Boulevard, Newark, California at 2:00 p.m., local time, or at any postponements or adjournments thereof, all of the shares of
Common Stock, par value $.01 per share, of StemCells, Inc. that the undersigned would be entitled to vote if personally present. The undersigned instructs such proxies or their substitutes to act on the following matter as specified by the
undersigned, and to vote in such manner as they may determine on any other matter that may properly come before the meeting.
WHEN PROPERLY EXECUTED, THIS PROXY
WILL BE VOTED AS DESIGNATED BY THE UNDERSIGNED. IF NO CHOICE IS SPECIFIED, THE PROXY WILL BE VOTED FOR EACH OF THE NOMINEES FOR DIRECTOR LISTED BELOW AND IN FAVOR OF PROPOSALS 2, 3, 4 AND 5. IN THIER DISCRETION, THE PROXIES ARE ALSO AUTHORIZED TO
VOTE UPON SUCH OTHER MATTERS AS MAY PROPERLY COME BEFORE THE MEETING AND ADJOURNMENT THEREOF.
Address change/comments:
(If you noted any Address Changes and/or Comments above, please mark corresponding box on the reverse side.)
0000284697_2 R1.0.1.25
Continued and to be signed on reverse side
Microbot Medical (NASDAQ:MBOT)
Historical Stock Chart
From Mar 2024 to Apr 2024
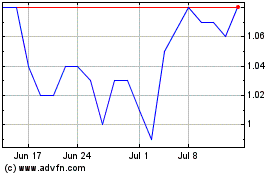
Microbot Medical (NASDAQ:MBOT)
Historical Stock Chart
From Apr 2023 to Apr 2024